Telangana TSBIE TS Inter 2nd Year Chemistry Study Material Lesson 6(b) Group-16 Elements Textbook Questions and Answers.
TS Inter 2nd Year Chemistry Study Material Lesson 6(b) Group-16 Elements
Very Short Answer Questions (2 Marks)
Question 1.
Why is dioxygen a gas but sulphur a solid?
Answer:
Due to small size and high electronegativity oxygen forms multiple bond (=) and exists as O2 molecule. These O2 molecules are held together by weak van der Waals forces. Hence oxygen exists as gas.
Due to large size and less electronegativity Sulphur forms strong S – S bonds and exists as S8 molecule. Hence Sulphur exists as solid.
Question 2.
What happens when
a) KClO3 is heated with MnO2,
b) O2 is passed through KI solution.
Answer:
a) Oxygen is evolved.
2KClO3 → 2KCl + 3O2
b) Iodide is oxidised to I2.
2KI + H2O + O3 → 2K0H + I2 + O2
Question 3.
Give two examples each for amphoteric oxides and neutral oxides.
Answer:
Examples of amphoteric oxides are Al2O3 and ZnO. Examples of neutral oxides are CO, NO and N2O.
![]()
Question 4.
Oxygen generally exhibits an oxidation state of – 2 only while the other members of the group show oxidation states of +2, + 4 and + 6 also – Explain.
Answer:
The multiple oxidation states in the case of elements other than oxygen are due to the availability of d-orbitals in the valence shell of the atoms.
Oxygen exhibits -2 oxidation state only generally except with F, where it shows + I in F2O2 and + II in F2O.
Question 5.
Write any two compounds in which oxygen shows an oxidation state different from -2. Give the oxidation states of oxygen in them.
Answer:
- In peroxides oxidation state of oxygen is – 1. Ex: H2O2
- In super oxides oxidation state of oxygen is -1/2. Ex : KO2
- In F2O, oxidation state of oxygen is + 2.
- In F2O2, oxidation state of oxygen is + 1.
Question 6.
Oxygen molecule has the formula O2 while sulphur has S8 – explain.
Answer:
Due to small atomic size and high electro-negativity in oxygen molecule, each oxygen atom is linked to other oxygen atom by a double bond. Hence its formula is O2.
\(: \ddot{\mathrm{O}}=\ddot{\mathrm{O}}:\)
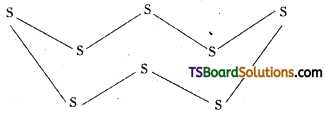
Due to large atomic size and less electro-negativity in sulphur molecule, eight ‘S’ atoms are linked together by single covalent bonds forming.
Puckered S8 rings with crown configuration. Hence formula of sulphur is S8.
Question 7.
Why is H2O a liquid while H2S is a gas? [IPE ’14]
Answer:
The O-H bond in H2O is highly polar. There are hydrogen bonds among the molecules of H2O. Hence it is present as a liquid.
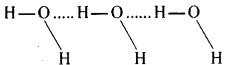
There are no hydrogen bonds among H2S molecules. So it exists as a gas at room temperature.
![]()
Question 8.
H2O is neutral while H2S is acidic-explain.
Answer:
Bond dissociation enthalpy of H-S bond is less than H – O bond. Hence H2S is acidic.
Question 9.
Name the most abundant element present in earth’s crust.
Answer:
Oxygen
Question 10.
Which element of group-16 shows highest catenation?
Answer:
Sulphur
Question 11.
Among the hydrides of chalcogens, which is most acidic and which is most stable?
Answer:
- Most acidic hydride of chalcogens is H2Te.
- Most stable hydride of chalcogens is H2O.
Question 12.
Give the hybridisation of sulphur in the following.
a) SO2
b) SO3
c) SF4
d) SF6
Answer:
Hybridisation of S in SO2 is sp2.
Hybridisation of S in SO3 is sp2.
Hybridisation of S in SF4 is sp3d.
Hybridisation of S in SF2 is sp3d2.
![]()
Question 13.
Write the names and formulae of any two oxyacids of sulphur. Indicate the oxidation state of sulphur in them.
Answer:
- Sulphurous acid = H2SO3
Oxidation state of sulphur is + 4. - Sulphuric acid = H2SO4
Oxidation state of sulphur is + 6. - Pyrosulphuric acid = H2S2O7
Oxidation state of sulphur is + 6.
Question 14.
Explain the structures of SF4 and SF6.
Answer:
Structure of SF4 :
Excited state configuration of S is
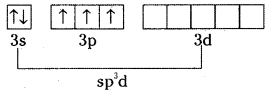
Hybridisation of Sulphur is sp3d.
SF4 has distorted trigonal bipyramidal structure with one orbital being occupied by a lone pair of electrons.
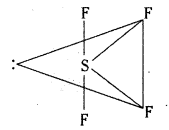
Structure of SF6:
Excited state configuration of Sulphur is
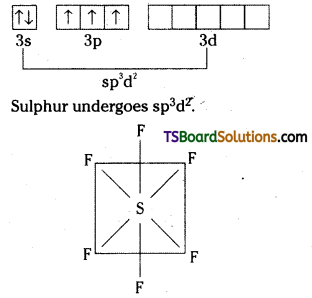
SF6 has octahedral symmetry.
Question 15.
Give one example each for
a) a neutral oxide
b) a peroxide
c) a super oxide
Answer:
a) Nitric oxide NO is a neutral oxide.
b) Na2O2 is a peroxide. Peroxides contain O – O bond.
c) KO2 is potassium super oxide.
![]()
Question 16.
What is tailing of mercury? How is it removed ? [AP & TS ’15]
Answer:
Ozone reacts with mercury to form Hg2O. Due to dissolution of Hg2O in Hg mercury loses its meniscus and sticks to the sides of glass. This is called tailing of mercury.
The menisus can be regained by shaking with water which dissolves Hg2O.
Question 17.
Write the principle involved in the quantitative estimation of ozone gas.
Answer:
Ozone liberates I2 from KI solution which can be titrated against a standard solution of Hypo using starch as an indicator.
2KI + H2O + O3 → 2KOH + I2 + O2
2Na2S2O3 + I2→ Na2S4O6 + 2NaI
Question 18.
Write the structure of Ozone.
Answer:
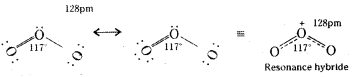
O-O bond lengths in ozone are identical (128pm) and molecule is angular with a bond angle of 117°.
Question 19.
SO2 can be used as anti-chlor. Explain.
Answer:
Sulphur dioxide reacts with chlorine in presence of charcoal to give sulphuryl chloride.
SO2(g) + Cl2 (g) → SO2Cl2 (l)
So it can remove chlorine and can be used as anti-chlor.
![]()
Question 20.
How is ozone detected?
Answer:
- Ozone turns startch iodide paper blue.
- It tails mercury.
Question 21.
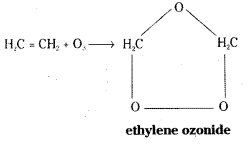
How does ozone react with ethylene? [Mar. 18 – A.P.]
Answer:
When ozone is bubbled through the solution of ethylene in an inert solvent like CCl4, at 195K, ethylene ozonide is formed.
NO + O3 → NO2 + O2
Question 22.
Out of O2 and O3, which is paramagnetic?
Answer:
O2 is paramagnetic. It contains two unpaired electrons in its molecular form O2.
Question 23.
Between O3 and O2, ozone is a better oxidising agent – why?
Answer:
Due to the ease with which it liberates atoms of nascent oxygen O3 → O2 + (O). Ozone acts as a powerful oxidising agent.
![]()
Question 24.
Write any two uses each for O3 and H3SO4.
Answer:
Uses of Ozone :
- It is used as a germicide, disinfectant, and for sterilising water.
- It is used for bleaching oils, ivory, flour, starch, etc.
Uses of H2SO4 :
- H2SO4 is used in the manufacture of fertilisers e.g. ammonium sulphate, super phosphate.
- Petroleum refining
Question 25.
Which form of sulphur shows paramagnetism?
Answer:
In vapour state sulphur partly exists as S2 molecule which has two unpaired electrons in the antibonding π* orbitals. Hence exhibits paramagnetism.
Question 26.
How is the presence of SO2 detected?
Answer:
SO2 decolourises KMnO4 solution in acid medium.
5SO2 + 2\(\mathrm{MnO}_4^{-}\) + 2H2O → 5\(\mathrm{SO}_4^{2-}\) + 4H+ + 2Mn++
![]()
Question 27.
Why are group-16 elements called chal- cogens ?
Answer:
The elements Oxygen, Sulphur, Selenium, Tellurium of group 16- are collectively known as chalcogens meaning ‘ore forming’. Since many metals occur as oxides or sulphides in nature. The name is derived from the Greek word for brass. It indicates the association of sulphur and its congeners with copper.
Question 28.
Among chalcogens, which has highest electronegativity and which has highest electron gain enthalpy? ‘
Answer:
Oxygen has highest electronegativity (3.5 Pauling scale) among chalcogens. Sulphur has highest electron gain enthalpy among chalcogens.
Question 29.
Which hydride of group-16 has highest boiling point and weakest acidic character ?
Answer:
H2O (water)
Short Answer Questions (4 Marks)
Question 30.
Justify the placement of O, S, Se, Te and Po in the same group of the periodic table in terms of electronic configuration, oxida-tion states and hydride formation.
Answer:
Electronic configuration: All the elements of group 16 have six electrons in the outermost shell and have ns2np4 general electronic configuration.
Oxidation states:
General oxidation states of Group 16 elements are -2, +2, +4 and +6. Oxygen cannot show higher oxidation states +4, +6 because of non-availability of d-orbitals in the valence shell. Sulphur, Selenium, and Tellurium usually show +4 oxidation state in their compounds with oxygen and +6 with fluorine. The stability of +6 oxidation state decreases down the group and stability of +4 oxidation state increases. This is due to inert pair effect.
Hydride formation :
All the chalcogens form covalent hydrides of the formula H2E. (E = S, Se, Te, Po)
Thermal stability of the hydrides decreases from H2O to H2Po. This is due to an increase in E – H bond length.
Water is a liquid while others are gases. Water exists as associated liquid due to hydrogen bonding.
The acidic character in aqueous solution increases from H2O to H2Te. This is due to decrease in charge density on conjugate bases OH–, SH–, SeH–, TeH–.
All the hydrides except water possess reducing property. The reducing property increases from H2O to H2Po. This trend can be attributed to decrease in thermal stability of hydrides from H2O to H2Po.
Thus there is a regular gradation in properties of these elements. Hence the inclusion of these elements in the same group is justified.
![]()
Question 31.
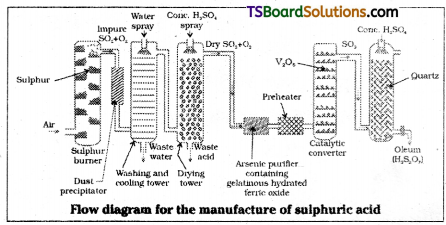
Describe the manufacture of H2SO4 by contact process. [TS ’16]
Answer:
Manufacture of H2SO4 by Contact process involves three steps.
i) Burning of sulphur or sulphide ores in air to generate SO2.
S + O2 → SO2
4FeS2 + 11O2 → 2Fe2O3 + 8SO2
ii) Conversion of SO2 to SO3 by the reaction with oxygen in the presence of a catalyst (V2O5).
![]()
iii) Absorption of SO3 in H2SO4 to give oleum.
SO3 + H2SO4 → H2S2O7
iv) Dilution of oleum with water gives sul-phuric acid.
H2S2O7 + H2O → 2H2SO4
Question 32.
How is ozone prepared? How does it react with the following ? [Mar. 19, 18, 17, AP]
a) PbS
b)KI
c) Hg
d) Ag
Answer:
Preparation of Ozone :
When a slow dry stream of oxygen is passed through a silent electrical discharge, oxygen is converted to ozone. This product is called ozonised oxygen.
3O2 → 2O3, ∆H° (298K) = +142 kJ/mol.
If high concentration of ozone (> 10%) is required, a battery of ozonisers can be used. Pure ozone can be condensed in a vessel surrounded by liquid oxygen.
Properties:
a) Pbs is oxidised to PbSO4.
PbS (s) + 4O3 (g) → PbSO4 + 4O2
b) I2 is liberated. I– is oxidised to I2.
2KI(aq) + H2O (l) + O3(g) → 2KOH(aq) + I2(s) + O3(g)
c) Hg2O formed sticks to the glass surface and mercury loses its meniscus due to dissolution of Hg2O in Hg. This is called tailing of mercury.
2Hg + O3 → Hg2O + O2
d) Silver metal is blackened. This is due to oxidation of the metal to its oxide
2Ag + O3 → Ag2O + O2 (oxidation)
![]()
Question 33.
Write a short note on the allotropy of sulphur.
Answer:
Sulphur forms numerous allotropes. The two common crystalline forms are α (alpha) or rhombic sulphur and β (beta) or monoclinic sulphur. The stable form at room temperature is rhombic sulphur. It transforms to monoclinic sulphur when heated above 369K.
Rhombic sulphur:
This is yellow in colour. It is formed by evaporating the solution of roll sulphur in CS2. It is soluble in CS2. m.p. 385.8K.
Monoclinic sulphur:
This form of sulphur is prepared by melting rhombic sulphur in a dish and cooling the solution till crust is formed. Two holes are made in the crust and remaining liquid poured out.
α – sulphur transforms to β – sulphur above 369K. At 369K both are stable. This temperature is called transition temperature.
Both forms have S8 puckered ring forms. It has a crown shape.
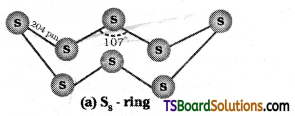
In cyclo – S6, the ring adopts chair form. At 1000K, S2 is the dominant species and is paramagnetic like O2.
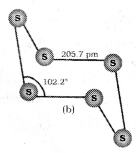
Question 34.
How does S02 react with the following?
a) Na2SO3 (aq)
b) Cl2
c) Fe3+ ions
d) KMnO4
Answer:
a) When SO2 is passed into sodium sulphite solution, sodium hydrogen sulphite is formed.
Na2SO3 + H2O + SO2 → 2NaHSO3
b) SO2 reacts with chlorine in the presence of charcoal to give sulphuryl chloride.
SO2(g) + Cl2(g) → SO2Cl2 (l)
c) SO2 reduces Fe3+ ions to Fe2+ ions.
2Fe3+ + SO2 + 2H2O → 2Fe2+ + SO42- + 4H+
d) It decolourises KMnO4.
5SO2 + 2MnO4– + 2H2O → 5SO42- + 4H+ + 2Mn++
![]()
Question 35.
Starting from elemental sulphur, how is H2S04 prepared?
Answer:
Manufacture of H2SO4 by Contact process involves three steps.
i) Burning of sulphur or sulphide ores in air to generate SO2.
S + O2 → SO2
4FeS2 + 11O2 → 2Fe2O3 + 8SO2
ii) Conversion of SO2 to SO3 by the reaction with oxygen in the presence of a catalyst (V2O5).
![]()
iii) Absorption of SO3 in H2SO4 to give oleum.
SO3 + H2SO4 → H2S2O7
iv) Dilution of oleum with water gives sul-phuric acid.
H2S2O7 + H2O → 2H2SO4
Question 36.
Describe the structures (shapes) of SO4-2 and SO3.
Answer:
Structure of SO3: SO3 has planar triangular shape in the gas phase. S is in sp2 hybridisation. Bond angle is 120°. S-O bond length 143pm,
In SO3, sulphur is in second excited state.
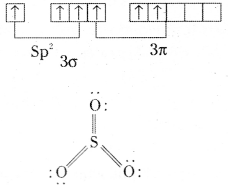
In the solid state it may be cyclic or chain.
Structure of Sulphite ion SO4– –
SO4– – is tetrahedral in shape. Bond angle is 109°28′: S – O bond length 149pm.
Sulphur undergoes sp3 hybridisation.
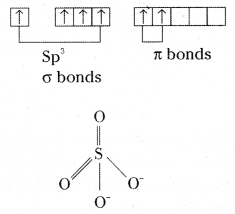
![]()
Question 37.
Which oxide of sulphur can act as both oxidising and reducing agent? Give one example each.
Answer:
SO2 acts both as oxidising and reducing agent.
- H2S is oxidised to sulphur.
SO2 + 2H2S → 2H2O + 3S - Fe3+ is reduced to Fe2+
2Fe3+ + SO2 + 2H2O → 2Fe2+ + SO42- + 4H+
Question 38.
Explain the conditions favourable for the formation of SO3 from SO2 in the contact process of H2SO4.
Answer:
2SO2 (g) + O2(g) → 2SO3 (g); ∆H = -196 kj/mol
The reaction is exothermic and reversible. Forward reaction leads to a decrease in volume. Therefore, low temperature and high pressure are favourable for maximum yield.
In practice, the plant is operated at a pressure of 2 bar and a temperature of 720K.
Question 39.
Complete the following.
a) KCl + H2SO4(conc.) →
b) Sucrose ![]()
c) Cu + H2SO4(conc.) →
d) C + H2SO4 (conc.) →
Answer:
a) HCl gas evolves.
2KCl + H2SO4 → K2SO4 + 2HCl
b) It removes water from carbohydrates.
![]()
c) SO2 gas evolves. Cu is oxidised.
Cu . 2H2SO4 → CuSO4 + SO2 + 2H2O
d) C is oxidised to CO2.
C + 2H2SO4 →CO2 + 2SO2 + 2H2O
![]()
Question 40.
Which is used for drying ammonia?
Answer:
Ammonia is dried using quick lime CaO.
Question 41.
Why cone. H2SO4, P4O10 and anhydrous CaCl2 cannot be used for dry ammonia? (Hint: ammonia reacts with them forming (NH4)2SO4 : (NH4)3 PO4 and CaCl2. 8 NH3
Answer:
Ammonia forms (NH4)3 PO4 with P4O10
Ammonia reacts with CaCl2 forming CaCl2 8NH3. Hence they cannot be used for drying ammonia.
Long Answer Questions (8 Marks)
Question 42.
Explain in detail the manufacture of sulphuric acid by contact process. [Mar. 2018 – TS]
Answer:
Contact process consists of three stages,
i) Burning of suphur or sulphide ores in air to generate SO2.
S + O2 → SO2
4FeS2 + 11O2 → 2Fe2O3 + 8SO2
ii) SO2 is oxidised to SO3 in presence of V2O5.
![]()
∆H = 196 kJ, mol-1
iii) Absorption of SO3 in H2SO4 to give oleum.
SO3 + H2SO4 → H2S2O7
Low temperature and high pressure are favourable for high yield of SO3. In practice, a pressure of 2 bar and a temperature of 720K are employed.
The SO3 gas from the catalytic converter is absorbed in concentrated H2SO4 to produce oleum. Dilution of oleum with water gives H2SO4 of the desired concentration.
SO3 + H2SO4 → H2S2O7
H2S2O7 + H2O → 2H2SO4
Question 43.
How is ozone prepared from oxygen? Explain its reaction with [AP Mar. ’18, 17, 16; 1PE ‘ 13,’ 14]
a) C2H4
b) KI
c) Hg
d) PbS
Answer:
Preparation of Ozone : When a slow dry stream of oxygen is passed through a silent electrical discharge, oxygen is converted to ozone. This product is called ozonised oxygen.
3O2 → 2O3, ∆H° (298K) = +142 kJ/mol.
If high concentration of ozone (> 10%) is required, a battery of ozonisers can be used. Pure ozone can be condensed in a vessel surrounded by liquid oxygen.
![]()
a) Ethylene reacts with ozone to form ethylene ozonide.
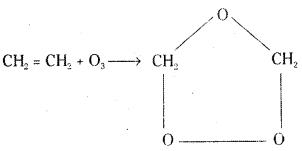
b) Iodide is oxidised to Iodine.
2KI + H2O + O3 → 2KOH + I2 + O2
c) Mercury is oxidised to Hg2O by ozone.
As Hg2O dissolves in Hg, Hg loses its meniscus and sticks to glass surface. It is called ‘tailing of mercury’.
2Hg + O3 → Hg2O + O2
d) PbS is oxidised to PbSO4 by O3.
PbS + 4O3 → PbSO4 + 4O2
Intext Questions – Answers
Question 1.
List the important sources of sulphur.
Answer:
- Sulphur exists primarily such as gypsum CaSO4 . 2H2O.
- Epsom salt MgSO4 . 7H2O
- Sulphides such as
galena PbS
zinc blende ZnS - eggs, proteins, garlic, onion ……………
Question 2.
Write the order of thermal stability of the hydrides of Group 16 elements
Answer:
The thermal stability of hydrides of Group -16 decreases from H2O to H2PO.
![]()
Question 3.
Why is H2O a liquid and H2S a gas at room temperature and pressure ?
Answer:
There is molecular association through hydrogen bonds in H2O. But, there is no such molecular association through hydrogen bond in H2S. H and H2O exists as liquid while H2S exists as gas.
Question 4.
Which of the following does not react with oxygen directly ? Zn, Ti, Pt, Fe
Answer:
Platinum.
Question 5.
Complete the following reactions :
i) C2H4 + O2 →
ii) 4 Al + 3O2 →
Answer:
i) C2H4 + 3O2 → 2CO2 + 2H2O
ii) 4 Al + 3O2 → 2 Al2O3
Question 6.
Why does O3 act as a powerful oxidizing agent?
Answer:
O3 can easily release nascent oxygen. Hence, it acts as powerful oxidizing agent.
![]()
Question 7.
How is O3 estimated quantitatively ?
Answer:
O3 reacts with excess of Kl solution and liberates Iodine. The liberated Iodine is titrated against a standard solution of sodium Thiosulphate.
Question 8.
What happens when SO2 is passed through an aqueous solution of Fe(III) salt ?
Answer:
It reduces Fe(lII) salts to Fe(II) salts.
2Fe+3 + SO2 + 2H2O → 2Fe+2 + SO4-2 + 4H+
Question 9.
Comment on the nature of two S-O bonds formed in S02 molecule. Are the two S-O bonds in this molecule equal ?
Answer:
The two S-O bonds in SO2 molecule are covalent and they are not equal. One oxygen is linked by single bond and the other oxygen is linked by double bond. The S-O bond length is larger than S = O bond length.
Question 10.
How is the presence of SO2 detected ?
Answer:
Due to the strong pungent smell, the presence of SO2 can be detected. It decolourises acidified KMNO4 solution.
![]()
Question 11.
Mention three areas in which H2SO4 plays an important role.
Answer:
- H2SO4 is used for manufacture of fertilisers. e.g.: ammoium sulphate
- Petroleum refining
- Detergent industry
- Metallurgical applications e.g.: electro-plating and galvanising.
Question 12.
Write the conditions to maximise the yield of H2SO4 by Contact process ?
Answer:
The yield of H2SO4 can be maximised by maintaining the following conditions.
a) low temperatures (720 k)
b) high pressures (2 bar).
Question 13.
Why is Ka2 < < Ka1 for in water?
Answer:
H2SO4 is a very strong acid in water largely because of its first ionisation to H3O+ and \(\mathrm{HSO}_4^{-}\). The ionisation of \(\mathrm{HSO}_4^{-}\) to H3O+ and SO42- is very very small.
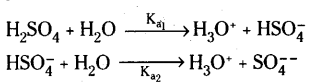
Hence , Ka2 < < Ka1