Telangana TSBIE TS Inter 2nd Year Chemistry Study Material 2nd Lesson Solutions Textbook Questions and Answers.
TS Inter 2nd Year Chemistry Study Material 2nd Lesson Solutions
Very Short Answer Questions (2 Marks)
Question 1.
Define the term solution.
Answer:
Homogeneous mixture of two or more than two components is called solution.
Question 2.
Define molarity.
Answer:
Molarity (M) is defined as number of moles of solute dissolved in one litre of (or one cubic decimetre) solution.
![]()
![]()
Question 3.
Define molality.
Answer:
Molality (m) is defined as the number of moles of the solute present in one kilogram (kg) of the solvent.
![]()
Question 4.
Give an example of a solid solution in which the solute is solid.
Answer:
In solid solution solute is solid. Here sol-vent is also solid. e.g., copper dissolved in gold (alloys).
Question 5.
Define mole fraction. (Mar. 2018 TS)
Answer:
Mole fraction is the ratio of number of moles of one component to the total number of moles of all the components in the solution.
Mole fraction of a component

Question 6.
Define mass percentage solution.
Answer:
The mass percentage of a component of a solution is defined as
Mass % of a component
Mass % of the component
![]()
Question 7.
What is ppm of a solution ?
Answer:
Parts per million
Number of parts of the
![]()
This method is convenient when a solute is present in trace amounts.
Question 8.
What role do the molecular interactions play in a solution of alcohol and water ?
Answer:
The interaction between alcohol and alcohol is hydrogen bonding and H2O and H2O is also hydrogen bonding. After mixing the interaction between alcohol and H2O is also hydrogen bonding but less than the hydrogen bonding in alcohol and H2O separately. So after mixing the interaction between alcohol and water decreases and the vapour pressure of solution increases forming low boiling point azeotrope.
Question 9.
State Raoult’s law. (AP & TS 16) (IPE 14) (Mar. 2018 . AP & TS)
Answer:
For any solution the partial vapour pressure of each volatile component in the solution is directly proportional to its mole fraction.
Question 10.
State Henry’s law.
Answer:
The solubility of a gas in a liquid is directly proportional to the partial pressure of the gas present above the surface of liquid or solution.
(or)
Mole fraction of gas in the solution is proportional to the partial pressure of the gas over the solution.
(or)
The partial pressure of the gas in vapour phase (p) is proportional to the mole fraction of the gas (x) in the solution.
P = KH × x
KH is the Henry’s law constant.
Question 11.
What is Ebullioscopic constant?
Answer:
The elevation in the boiling point of one molal solution i.e., when one mole of solute is dissolved in 1 kg of solvent, is called boiling point elevation constant or molal elevation constant or ebullioscopic constant.
ΔTb = Kb m.
unit of Kb = K kg mol-1
Question 12.
What is Cryoscopic constant?
Answer:
The depression in freezing point of one molal solution i.e., when one mole of solute is dissolved in 1 kg of solvent, is called freezing point depression constant or molal depression constant or cryoscopic constant.
ΔTf = Kf m.
unit of Kf = K kg mol-1
![]()
Question 13.
Define Osmotic pressure. (AP 16, 15) (Mar. 2018 – AP)
Answer:
When a solution is separated from a solvent by a semipermeable membrane or if a dilute solution is separated from concentrated solution by a semipermeable membrane, the pressure that just prevents passage of solvent into solution or solvent from dilute solution into concentrated solution is called osmotic pressure.
(or)
The extra pressure that is to be applied on the solution side when the solution and solvent are separated by a semipermeable membrane to stop osmosis.
Question 14.
What are isotonic solutions ? TS Mar. 19; (AP 17, 15; TS 16)
Answer:
Two solutions having same osmotic pressure at a given temperature are called isotonic solutions.
Question 15.
Amongst the following compounds, identify which are insoluble, partially soluble and highly soluble in water,
(i) Phenol
(ii) Toluene
(iii) Formic acid
(iv) Ethylene glycol
(v) Chloroform
(vi) Pentanol.
Answer:
Insoluble : Chloroform, toluene.
Partially soluble : Phenol, pentanol.
Highly soluble: Formic acid, ethylene glycol.
Question 16.
Calculate the mass percentage of aspirin (C9H8O4) in acetonitrile (CH3CN) when 6.5 gm of C9H8O4 is dissolved in 450g of CH3CN.
Answer:
Mass percentage of aspirin.
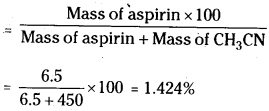
Question 17.
Calculate the amount of benzoic acid (C6H5COOH) required for preparing 250ml of 0.15M solution in methanol.
Answer:
Molecular mass of benzoic acid (C6H5COOH) = 12 × 6 + 5 + 12 + 32 + 1 = 122 g mol-1
moles of benzoic = M × V
= 250 × 0.15 = 37.5
= 37.5 × 10-3 mol
Amount of benzoic acid = Moles × MW2
= 37.5 × 10-3 × 122
= 4.575 g.
Question 18.
The depression in freezing point of water observed for the same amount of acetic acid, dichloro – acetic acid and trichloro acetic acid increases in the order given above. Explain briefly.
Answer:
Increasing order of ΔTf (depression in freezing point) is Acetic acid < dichloroacetic acid < trichloroacetic acid.
Due to more electronegativity of Cl atom it exerts -1 (inductive effect) consequently dichloroacetic acid is the stronger acid than acetic acid. As the number of chlorine atoms increases the inductive effect also increases. Hence trichloro acetic acid is stronger acid than dichloroacetic acid.
Question 19.
What is van’t Hoffs factor ‘i’ and how is it related to ‘α’ in the case of a binary ele-ctrolyte (1 : 1) ?
Answer:
van’t Hoff introduced a factor ‘i’ known as the van’t Hoff factor to account the extent of dissociation or association. The factor ‘i’ is defined as

If α represents the degree of association of the solute for an equilibrium.
nA \(\rightleftharpoons\) An
Total number of particles at equilibrium is 1 – α + \(\frac{\alpha}{2}\) = 1 – \(\frac{\alpha}{2}\)
Then i = \(\frac{\alpha / 2}{1 \cdot 1}\) = \(\frac{\alpha}{2}\)
For dissociation
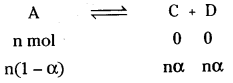
Total moles of particles aren(1 – α + α + α)
= n(1 + α)
i = \(\frac{n(1+\alpha)}{n}\) = 1 + α
![]()
Question 20.
What is relative lowering of vapour pressure ? (AP Mar. 19)
Answer:
The ratio of the lowering of vapour pressure of a solution containing a non – volatile solute to the vapour pressure of pure solvent is called relative lowering of vapour pressure.
It can be shown as R.L.V.P. = \(\frac{p^{\circ}-p}{p^{\circ}}\)
Here p° is the vapour pressure of pure solvent, p is the vapour pressure of solution containing non – volatile solute.
Question 21.
Calculate the mole fraction of H2SO4 in a solution containing 98% H2SO4 by mass. (Mar. 2018 – TS)(IPE 14)
Answer:
Mass of water = 2 gm.
Moles of water = \(\frac{2}{18}\) = \(\frac{1}{9}\) (M.Wt. of H2O = 18)
Mass of H2SO4 = 98 gm.
Moles of H2SO4 = \(\frac{98}{98}\)
= 1 (Mol. Wt. of H2SO4 = 98)
Moles fraction of H2SO4 = \(\frac{1}{1+\frac{1}{9}}\) = \(\frac{9}{10}\) = 0.9
Question 22.
How many types of solutions are formed ? Give an example for each type of solution.
Answer:
Depending on the type of solvent three types of solutions will be formed.
| Type of Solution | Solute | Solvent | Common Examples |
| Gaseous Solutions | Gas Liquid Solid | Gas Gas Gas | Mixture of oxygen and nitrogen gases Chloroform mixed with nitrogen gas Camphor in nitrogen gas. |
| Liquid Solutions | Gas Liquid Solid | Liquid Liquid Liquid | Oxygen dissolved in water. Ethanol dissolved in water. Glucose dissolved in water. |
| Solid Solutions | Gas Liquid Solid | Solid Solid Solid | Solutions of hydrogen in palladium. Amalgam of mercury with sodium. Copper dissolved in gold. |
Question 23.
Define mass percentage, volume percentage and mass to volume percentage solutions.
Answer:
Mass percentage (w/w) : The mass percentage of a component of a solution is defined as
Mass % of a component
Mass of the component

Volume percentage (V/V) : The volume percentage is defined as
Volume % of a component
![]()
Mass to volume percentage (w/v): It is the mass of solute dissolved in 100 ml of the solution.
Question 24.
Concentrated nitric acid used in the laboratory work is 68% nitric acid by in aqueous solution. What should be the molarity of such a sample of the acid if the density of the solution is 1.504 g mL-1?
Answer:
68% by mass implies that 68 g of HNO3 is present in 100 g of solution.

Question 25.
A solution of glucose in water is labelled as 10% w/w. What would be the molarity of the solution?
Answer:
Wt. of glucose = 1o g
Wt. of solution = 100 gm
Wt. of water = 100 – 10 = 90 gm.
Considering density of water as 1 gm mL-1
Volume of water = 90 mL
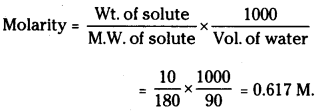
Question 26.
A solution of sucrose In water is labelled as 20% w/w. What would be the mole fraction of each component in the solution?
Answer:
Wt. of sucrose = 20
Moles of sucrose = \(\frac{20}{342}\) = 0.0585
Wt. of solution = 100 g
Wt. of water = 100 – 20 = 80 g
Moles of water = \(\frac{80}{18}\) = 4.44
Mole fraction of sucrose = \(\frac{0.0585}{0.0585+4.44}\)
= 0.013
Mole fraction of water = \(\frac{4.44}{0.0585+4.44}\)
= 0.987
![]()
Question 27.
How many ml of 0.1 MHCl is required to react completely with 1.0 g mixture of Na2CO3 and NaHCO3 containing equimolar amounts of both ?
Answer:
Let Na2CO3 be x g
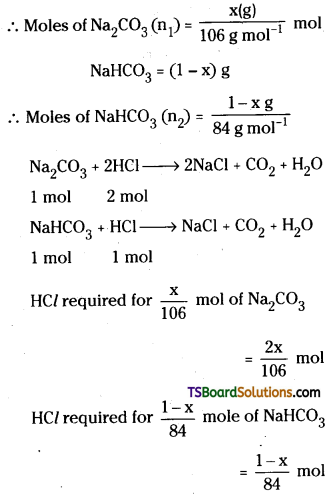
Since, given mixture contains equimolar amount.
∴ \(\frac{\mathrm{x}}{106}\) = \(\frac{1-x}{84}\) (or) x = 0.56
Total HCl required
= \(\left[\frac{2 x}{106}+\frac{1-x}{84}\right]\) = \(\frac{2 \times 0.56}{106}\) + \(\frac{1-0.56}{84}\)
= 0.01578 mol
If V is the volume of HCl required, then
V(L) × 0.1 = 0.01578
(or) V(L) = 0.1578L (or) 157.8 mL.
Question 28.
A solution is obtained by mixing 300g of 25% solution and 400g of 40% solution by mass. Calculate the mass percentage of the resulting solution.
Answer:
Mass of solute in 300 g of 25% solution.
= \(\frac{300 \times 25}{100}\) = 75 g
Mass of solute in 400 g of 40% solution
= \(\frac{400 \times 40}{100}\) = 160 g
Total mass of solute = 75 + 160 = 235 g
Total mass of solution = 700 g
% solute in final solution = \(\frac{235 \times 100}{700}\)
= 33.5%
% of water in final solution = 100 – 33.5
= 66.5%
Question 29.
An antifreeze solution is prepared from 222.6g of ethylene glycol (C6H6O2) and 200g of water (solvent). Calculate the molality of the solution.
Answer:
Mass of glycol (w1) = 222.6 g
Moles of glycol (n1)
= ![]()
= \(\frac{222.6}{62}\)
= 3.59 mol
Mass of water (w2) = 200 g = \(\frac{200 \times 1 \mathrm{~kg}}{1000}\)
= 0.2 kg
Mass of solution (w1 + w2) = 200 + 222.6
= 422.6 g
Density of solution (d) = 1.072 g mL-1
Volume of solution (V)
= ![]()
= \(\frac{422.6}{1.072}\)
= 394.22 mL (or) 0.3942 L
Molality (m) = \(\frac{\mathrm{n}_1}{\mathrm{w}_2 \mathrm{~kg}}\) = \(\frac{3.59 \mathrm{~mol}}{0.2 \mathrm{~kg}}\)
= 17.95 mol kg
Molarity (M) = \(\frac{n_1}{V \text { in lit }}\) = \(\frac{3.59}{0.3942}\) = 9.1 mol L-1
Question 30.
Why do gases always tend to be less soluble in liquids as the temperature is raised ?
Answer:
Dissolution of gases is exothermic process. It is because dissolution of a gas in a liquid decreases the entropy (ΔS < 0). Thus increase of temperature tends to push the equilibrium (Gas + solvent \(\rightleftharpoons\) solution, ΔH = -Ve) in the backward direction, thereby, suppressing the dissolution.
![]()
Question 31.
What is meant by positive deviations from Raoult’s law and how is the sign of Δmix H related to positive deviation from Raoult’s law ?
Answer:
The solutions which do not obey Raoult’s law and are accompanied by change in enthalpy and change in volume during their formation are called non – ideal solutions.
In the solutions showing positive deviations the partial vapour pressure of each component (say A and B) of solution is greater them the vapour pressure as expected according to Raoult’s law. In this type of solutions the solvent – solvent and solute – solute interactions are stronger than solvent – solute interactions since in the solution, the interactions among molecules become weaker, their escaping tendency increases which results in the increase in their partial vapour pressures. In such solutions total vapour pressure of the solution is also greater than the vapour pressure required according to the Raoult’s law.
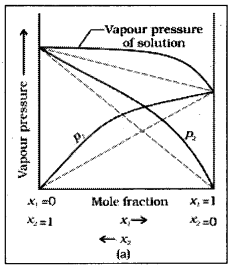
For this type of non – ideal solutions exhibiting positive deviations.
- PA < \(P_A^0 x_A\); PB > \(\mathrm{P}_{\mathrm{B}}^0 \mathrm{x}_{\mathrm{B}}\)
- ΔmixH = +ve
- ΔmixV = +ve
Question 32.
What is meant by negative deviation from Raoult’s law and how is the sign of ΔmixH related to negative deviation from Raoult’s law?
Answer:
The solutions which do not obey Raoult’s law and are accompanied by change in enthalpy and change in volume during their formation are called non – Ideal solutions. In the solutions showing negative deviations the partial vapour pressure of each component of solution is less than the vapour pressure as expected according to Raoult’s law. In this type of solutions solvent – solvent and solute – solute interactiòns are weaker than that of solvent – solute interactions.
So the interactions among molecules in solution become stronger. Hence the escaping tendency of molecules decrease which results in the decrease in their partial vapour pressure. In such solutions total vapour pressure of the solution is also less than the vapour pressure expected according to Raoult’s law.
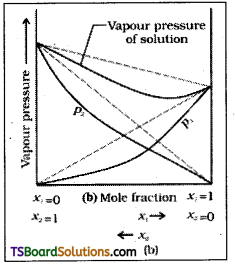
For these solutions exhibiting negative deviations
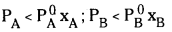
- Δmix H = -ve
- ΔmixV = -ve
Question 33.
The vapour pressure of water is 12.3 k Pa at 300K. Calculate the vapour pressure of 1 molar solution of a non – volatile solute in it.
Answer:
Vapour pressure of water \(\mathrm{P}_{\mathrm{H}_2}^0 \mathrm{O}\) 12.3 k Pa
In 1 molar solution
Moles of water nH2O = \(\frac{1000}{18}\) = 55.5 mol
Moles of solute nB = 1 mol
Mole fraction of H2O (XH2O) = \(\frac{\mathrm{n}_{\mathrm{H}_2 \mathrm{O}}}{\mathrm{n}_{\mathrm{H}_2 \mathrm{O}}+\mathrm{n}_{\mathrm{B}}}\)
= \(\frac{55.5}{55.5+1}\) = 0.982
![]()
Question 34.
Calculate the mass of a non – volatile solute (molar mass 40g mol-1) which should be dissolved in 114g Octane to reduce its vapour pressure to 80%. (TS 16; IF’ 14)
Answer:
Vapour pressure of solution (p) = 80% of \(\mathrm{P}_{\mathrm{A}}^0\) = 0.8\(\mathrm{P}_{\mathrm{A}}^0\)
Let the mass of solute be w g
∴ Moles of solute nB = \(\frac{\mathrm{W}_{\mathrm{B}}}{\mathrm{M}_{\mathrm{B}}}\) = \(\frac{W}{40}\) mol
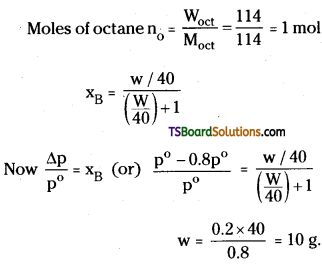
Question 35.
A 5% solution (by mass) of cane sugar in water has freezing point of 271K. Calculate the freezing point of 5% glucose in water If freezing point of water is 273.15K.
Answer:
Molarity of sugar solution (m) = \(\frac{\mathrm{W}_{\mathrm{B}} \times 1000}{\mathrm{M}_{\mathrm{B}} \times \mathrm{W}_{\mathrm{A}}}\)
(or) m = \(\frac{5 \mathrm{~g} \times 1000 \mathrm{~g} \mathrm{~kg}^{-1}}{342 \mathrm{~g} \mathrm{~mol}^{-1} \times 95 \mathrm{~g}}\) = 0.154 mol kg-1
ΔTf = 273.15 – 271 = 2.15°
Now ΔTf = Kf × m or Kf = \(\frac{\Delta \mathrm{T}_{\mathrm{f}}}{\mathrm{m}}\) = \(\frac{2.15}{0.154}\) = 13.96
Molailty of glucose solution (m)
= \(\frac{5 \mathrm{~g} \times 1000 \mathrm{~g} \mathrm{~kg}^{-1}}{180 \mathrm{~g} \mathrm{~mol}^{-1} \times 95 \mathrm{~g}}\) = 0.292 mol kg-1
ΔTf = Kfm= 13.96 × 0.292 = 4.08°
Freezing point of glucose solution
= 273.15 – 4.08 = 269.07 K.
Question 36.
If the osmotic pressure of glucose solution is 1.52 bar at 300K. What would be its concentration if R = 0.083L bar mol-1 K-1?
Answer:
Temperature T = 300 K
Osmotic pressure = 1.52 bar
R = 0.083 L bar mol-1 K-1
π = CRT (or) C = \(\frac{\pi}{\mathrm{RT}}\)
= \(\frac{1.52 \mathrm{bar}}{0.083 \mathrm{~L} \mathrm{bar} \mathrm{mol}^{-1} \mathrm{~K}^{-1} \times 300}\)
∴ = 0.061 mol L-1
Question 37.
Vapour pressure of water at 293K is 17.535mm Hg. Calculate the vapour pressure of the solution at 293K when 25g of glucose is dissolved in 450 g of water. (AP Mar. 19)
Answer:
Vapour pressure of water \(\mathrm{p}_{\mathrm{H}_2 \mathrm{O}}^{\mathrm{O}}\) = 17.535 mm.
Let vapour pressure of solution be ps

Question 38.
How is molar mass related to the elevation in boiling point of a solution ?
Answer:
To calculate the molar mass of an unknown non – volatile compound a known mass (say WBg) of it is dissolved in a known mass (say WA g) of some suitable solvent and elevation in its boiling point (ΔTb) is determined. Let MB be the molar mass of the compound. Then
Molarity of solution m = \(\frac{W_B}{M_B} \times \frac{1000}{W_A}\)
We know, ΔTb = Kb × m = Kb × \(\frac{W_B}{W_A}\) × \(\frac{1000}{M_B}\)
MB = \(\frac{\mathrm{K}_{\mathrm{b}} \times \mathrm{W}_{\mathrm{b}} \times 1000}{\mathrm{~W}_{\mathrm{A}} \times \Delta \mathrm{T}_{\mathrm{b}}}\)
Knowing Kb, WB, WA and the molar mass of the compound can be calculated
from the above relation. This method is known as ebullioscopic method.
Question 39.
What is an Ideal solution?
Answer:
An ideal solution may be defined as the solution which obeys Raoults law over the entire range of concentration and temperature.
According to Raoult’s law, the vapour pressure of a volatile component (pA) of the solutions is equal to the product of its mole fraction (xA) in solution and vapour pressure in pure state \(\mathrm{P}_{\mathrm{A}}^0\).
PA = \(\mathrm{p}_{\mathrm{A}}^0 \mathrm{x}_{\mathrm{A}}\)
The formation of ideal solution neither involve any change in enthalpy nor in volume. An ideal solution,
- should obey Raoults law i.e., pA = \(p_A^0 x_A\) and pB = \(p_B^0 x_B\)
- ΔmixH = 0
- ΔmixV = 0
In ideal solutions the solvent – solvent and solute – solute interactions are almost the same type as solvent – solute interactions.
![]()
Question 40.
What is relative lowering of vapour pressure ? How is it useful to determine the molar mass of a solute ?
Answer:
When a non – volatile solute such as urea, glucose etc., is dissolved in a volatile solvent such as water, the vapour pressure of solution will be less than that of pure solvent. This is known as lowering of vapour pressure.
The ratio of lowering of vapour pressure to that vapour pressure of pure solvent is known as relative lowering of vapour pressure.
\(\frac{\Delta \mathrm{p}}{\mathrm{p}_{\mathrm{A}}^0}\) = Relative lowering of vapour pressure
Δp = \(\mathrm{p}_{\mathrm{A}}^0-\mathrm{p}_{\mathrm{A}}\) = lowering of vapour pressure
\(\mathrm{p}_{\mathrm{A}}^0\) = vapour pressure of pure solvent
Determination of molar mass of a solute:
According to Raoult’s law the relative lowering of vapour pressure i equal to the mole fraction of the solute.
\(\frac{\Delta \mathrm{p}}{\mathrm{p}_{\mathrm{A}}^0}\) = xB
xB = mole fraction of solute,
If a known mass (WB) of the solute is dissolved in a known mass (WA) of solvent to prepare a dilute solution and the relative lowering of vapour pressure Is determined experimentally the molar mass of solvent (MA) is known, the molar mass of solute MB can be determined as follows.
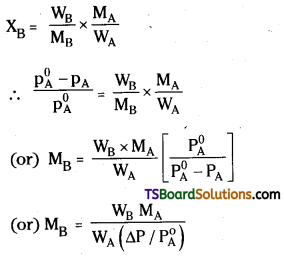
In this equation all the parameters except MB are known and hence MB can be calculated.
Question 41.
How is molar mass related to the depre-ssion in freezing point of a solution ?
Answer:
To determine the molar mass of an unknown non – volatile compound a known mass (say WBg) of it is dissolved in a known mass (say WAg) of some suitable solvent and depression in the freezing point (ΔTf) is determined. Let MB be the molar mass of the compound. Then
Molarity of the solution m = \(\frac{W_B}{M_B} \times \frac{1000}{W_A}\)
We know ΔTf = Kf × m = Kf × \(\frac{W_B}{W_A}\) × \(\frac{1000}{M_B}\)
MB = \(\frac{\mathrm{K}_{\mathrm{b}} \times \mathrm{W}_{\mathrm{B}} \times 1000}{\mathrm{~W}_{\mathrm{A}} \times \Delta \mathrm{T}_{\mathrm{f}}}\)
Knowing Kf, WB, WA and ΔTf, the molar mass of the compound can be calculated from the above relation. This method is called cryoscopic method.
Long Answer Questions (8 Marks)
Question 42.
An aqueous solution of 2% non – volatile solute exerts a pressure of 1.004 bar at the normal boiling point of the solvent. What is the molecular mass of the solute ?
Answer:
The vapour pressure of pure water
\(\mathbf{p}_{\mathrm{A}}^0\) = 1 atm = 1.013 bar
Vapour pressure of solution (p) = 1.004 bar
WB = 2g = WA + WB = 100g; WA = 98g
Now \(\frac{\mathbf{p}_{\mathrm{A}}^0-\mathrm{P}}{\mathbf{p}_{\mathrm{A}}^0}\) = \(\frac{\mathrm{n}_{\mathrm{B}}}{\mathrm{n}_{\mathrm{A}}}\) = \(\frac{W_B / M_B}{W_A / M_A}\)
or MB = \(\frac{W_B M_A}{W_A\left(\Delta P / P_A^0\right)}\) = \(\frac{2 \times 18 \times 1.013}{98 \times(1.013-1.004)}\)
= 41.35 g. mol-1
Question 43.
Heptane and Octane form an ideal solution. At 373K the vapour pressure of the two liquid components are 105.2 kPa and 46.8 kPa respectively. What will be the vapour pressure of a mixture of 26.0 g heptane and 35g of octane ?
Answer:
Moles of C7H16(nH) = \(\frac{W_{(H)}}{M_{(H)}}\) = \(\frac{25}{100}\) = 0.25 mol
Moles of C8H18 (nO) = \(\frac{W_{(0)}}{M_{(0)}}\) = \(\frac{35}{114}\) = 0.307 mol
Mole fraction of C7H16 = (XH) = \(\frac{0.25}{0.25+0.307}\)
= \(\frac{0.25}{0.557}\) = 0.466
Mole fraction of C8H18 (X0) = 1 – 0.449
= 0.534
Vapour pressure of heptane (pH)
\(\mathrm{p}_{\mathrm{H}}^{\mathrm{O}}\) × XH
= 105.2 \(\mathrm{Kp}_{\mathrm{a}}\) × 0.449
= 47.2348 \(\mathrm{Kp}_{\mathrm{a}}\) ≅ 49.02 \(\mathrm{kp}_{\mathrm{a}}\)
Vapour pressure of octane (PO)
= \(P_{\mathrm{H}}^O\) × XO
= 46.8 \(\mathrm{kp}_{\mathrm{a}}\) × 0.551 = 24.99 \(\mathrm{kp}_{\mathrm{a}}\)
Total vapour pressure PTotal = PH + PO
= 49.02 + 24.99 = 74.09 \(\mathrm{kp}_{\mathrm{a}}\)
Question 44.
A solution containing 30g of non – volatile solute exactly in 90g of water has a vapour pressure of 2.8 kPa at 298k. Further 18g of water is then added to the solution and the new vapour pressure becomes 2.9 \(\mathrm{kp}_{\mathrm{a}}\) at 298K. Calculate
(i) The molar mass of the solute and
(ii) Vapour pressure of water at 298k.
Answer:
Moles of solute (nB) = \(\frac{30}{M_B}\)
Moles of H2O(nH2O) = \(\frac{90}{18}\) = 5 mol
Mole fraction of H2O
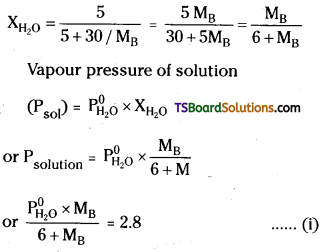
After adding 18g(= 1 mol) of water to solution new mole fraction of water(\(\mathrm{X}_{\mathrm{H}_2 \mathrm{O}}^{\prime}\)) is

Question 45.
Two elements A and B form compounds having formula AB2 and AB4. When dissolved in 20g of Benzene (C6H6), 1g of AB2 lowers the freezing point by 2.3K whereas 1.0g of AB4 lowers it by 1.3K. The molar depression constant for benzene is 5.1 K kg mol-1. Calculate atomic masses of A and B.
Answer:
We know

Let the atomic weight of A = x
Let the atomic weight of B = y
x + 2y = 110.86
x + 4y = 196.15
Solving for x and y, we get
y = 42.64, x = 25.58
∴ Atomic weight of A = 25.8 u
Atomic weight of B = 42.64 u
Question 46.
Calculate the depression In the freezing point of water when 10g of CH3CH2CHClCOOH is added to 250g of water. Ka = 1.4 × 10-3, Kf = 1.86K kg mol-1.
Answer:
Calculation of molarity of the solution:
Mass of the solution = 250 + 10 = 260 g

Calculation of van’t Hoffs factor (i) : Let degree of dissociation of acid be α since acid is monobasic acid, therefore α and Ka of acid are related as
α = \(\sqrt{K_{\mathrm{a}} / \mathrm{c}}\) = \(\sqrt{\frac{1.4 \times 10^{-3}}{0.284}}\) = 0.07
Van’t Hoff factor and degree of dissociation are related as
α = \(\frac{i-1}{m-1}\) = \(\frac{i-1}{2-1}\) or i = 1 + α = 1 + 0.07
(or) i = 0.7
Calculation of depression in freezing point ΔTf
ΔTf = i × Kf × m = \(\frac{1.07 \times 1.86 \times 10 \times 1000}{122.5 \times 250}\)
= 0.649 = 0.65°C
![]()
Question 47.
19.5 g of CH2FCOOH is dissolved in 500g of water. The depression in freezing point of water observed is 1.0°C. Calculate the van’t Hoff factor and dissociation constant of fluoroacetic acid.
Answer:
Mass of solution = 500 + 19.5 = 519.5 g.
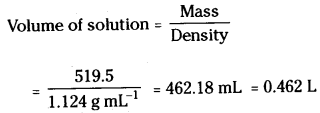
Moles of CH2F COOH (nB) = \(\frac{W_B}{M_B}\) = \(\frac{19.5}{78}\)
= 0.25 mol
Molarity of solution (M) = \(\frac{n_B}{V_{(L)}}\) = \(\frac{0.25}{0.462}\)
= 0.541 moL-1
Mass of water (WA) = 500 g = 0.5 kg
Molarity of solution (m) = \(\frac{\mathrm{n}_{\mathrm{B}}}{\mathrm{W}_{\mathrm{A}} \mathrm{kg}}\) = \(\frac{0.25 \mathrm{~mol}}{0.25 \mathrm{~kg}}\)
= 0.5 mol kg-1
ΔTf = i × Kf × m or i = \(\frac{\Delta \mathrm{T}_{\mathrm{b}}}{\mathrm{K}_{\mathrm{b}} \times \mathrm{m}}\)
(or) i = \(\frac{1(\mathrm{~K})}{1.86 \mathrm{k} \mathrm{kg} \mathrm{mol}^{-1} \times 0.5 \mathrm{~mol} \mathrm{~kg}^{-1}}\)
= 1.0753
Each molecule of CH2F COOH dissociate into 2 particles as
CH2F COOH \(\rightleftharpoons\) CH2F COO– + H+
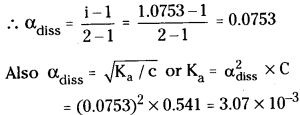
Question 48.
100g of liquid A(molar mass 140g mol-1) was dissolved in 1000g of liquid B(molar mass 180g mol-1). The vapour pressure of pure liquid B was found to be 500 torr. Calculate the vapour pressure of pure liquid A and its vapour pressure in the solution if the total vapour pressure of the solution is 475 torr.
Answer:
Let vapour pressure of pure A be \(\mathbf{p}_{\mathrm{A}}^0\)
\(p_{\mathrm{B}}^0\) = 500 torr ; nA = \(\frac{100}{140}\) = 0.714 mol
nB = \(\frac{1000}{180}\) = 5.55 mol
xA = \(\frac{0.714}{5.55+0.714}\) = 0.114
xB = 1 – 0.114 = 0.886
ptotal = \(p_A^0 x+p_B^0 x_B\)
(or) 475(torr) = \(p_A^0\) × 0.114 + 500 × 0.886
(or) \(\mathrm{p}_{\mathrm{A}}^0\) = 280.7 torr
PA = 280.70 × 0.114 = 32 Torr
Question 49.
Determine the amount of CaCl2 (i = 2.47) dissolved in 2.5 litre of water such that its osmotic pressure is 0.75 atm at 27°C.
Answer:
We know that π = CRT
or π = \(\frac{\mathrm{i} \times \mathrm{W}_{\mathrm{B}} \times \mathrm{R} \times \mathrm{T}}{\mathrm{M}_{\mathrm{B}} \times \mathrm{V}_{\mathrm{L}}}\)
or WB = \(\frac{\pi \times M_B \times V_L}{i \times R \times T}\)
van’t Hoff factor i = 2.47
Osmotic pressure p = 0.75 atm.
temperature T = 273 + 27 = 300 K.
Mol. wt of CaCl2, MB = 111
Volume of solution = 2.5 L
Constant R = 0.0821 L atm mol-1 K-1
WB = \(\frac{0.75 \times 111 \times 2.5}{2.7 \times 0.0821 \times 300}\) = 3.40 g
Moles of CaCl2 = \(\frac{3.4}{111}\) = 0.03 mol.
Question 50.
Determine the osmotic pressure of a solution prepared by dissolving 25 mg of K2SO4 in two litre of water at 25°C assuming that it is completely disassociated.
Answer:
K2SO4 is strong electrolyte, ionises completely
K2SO4 → 2K+ + \(\mathrm{SO}_4^{2-}\)
Therefore i = 3
Volume of the solution = 2L
Mass of K2SO4 (WB) = 25 mg = 0025 g
Molar mass of K2SO4 (MB) = 174 g mol-1
π = iCRT = i × \(\frac{3 \times 0.025 \times 0.0821 \times 298}{174 \times 2}\)
= 5.27 × 10-3 atm
Question 51.
Benzene and Toluene form ideal solution over the entire range of composition. The vapour pressure of pure benzene and toluene at 300K are 50.71 mm of Hg and 32.06 mm of Hg respectively. Calculate the mole fraction of benzene in vapour phase if 80g of benzene is mixed with 100g of toluene.
Answer:
If yb is the mole fraction of benzene in vapour form


Intext Questions – Answers
Question 1.
Calculate the mass percentage of benzene (C6H6) and carbon tetrachloride (CCl4) if 22g of benzene is dissolved in 122g of carbon tetrachloride.
Answer:
Weight of Benzene, w = 22 g
Weight of carbon tetrachloride, W = 122 g
Mass percentage of benzene = \(\frac{w \times 100}{W+w}\)
= \(\frac{22 \times 100}{22+122}\) = 15.27%
Mass percentage of carbon tetrachloride
= \(\frac{W \times 100}{W+w}\) = \(\frac{122 \times 100}{22+122}\) = 84.73%
Question 2.
Calculate the mole fraction of benzene in solution containing 30% by mass of it in carbon tetrachloride.
Answer:
Mass of benzene, w = 30 g
Mol. wt of benzene = 78 g mol-1
Moles of benzene = \(\frac{30}{78}\) = 0.385
Mass of CCl4 = 70 g
Mol. wt. of CCl4 = 154 g mol-1
Moles of CCl4 = \(\frac{70(\mathrm{~g})}{154(\mathrm{~g})}\) = 0.454
Mole fraction of benzene = \(\frac{0.385}{0.385+0.454}\)
Mole fraction of CCl4 = (1 – XBenzene)
= 1 – 0.459 = 0.541
Question 3.
Calculate the molarity of each of the following solutions
(a) 30 g of CO(NO3)2. 6H2O in 4.3L of solution
(b) 30 ml of 0.5 M H2SO4 diluted to 500 ml.
Answer:
a) Mass of CO (NO3)2. 6H20 = 30 g
Molar mass of CO(NO3)2. 6H2O
= 297 g mol-1.
Volume of solution = 4.3 L
Molarity M = \(\frac{30(\mathrm{~g})}{297 \mathrm{gmol}^{-1} \times 4.3 \mathrm{~L}}\)
= 0.023 mol L-1
b) Initial volume V1 = 30 ml
Molarity M1 = 0.5 mol L-1
Final volume V2 = 500 ml
Final molarity M2 =?
M1V1 = M2V2 (or) M2 = \(\frac{M_1 V_1}{V_2}\)
= \(\frac{0.5 \times 30}{500}\) = 0.03 mol L-1
Question 4.
Calculate the mass of urea (NH2CONH2) required in makIng 2.5 kg of 0.25 molal aqueous solution.
Answer:
The mass of urea required = WB
Molar mass of urea MB = 60
Mass of solution WA + WB = 2.5 kg = 2500 gm.
Mass of solvent WA = 2500 – WB
Molarity m = 0.25 mol kg-1
m = \(\frac{\mathrm{W}_{\mathrm{B}} \times 1000}{\mathrm{M}_{\mathrm{A}} \times \mathrm{W}_{\mathrm{B}}}\)
0.25 = \(\frac{\mathrm{W}_{\mathrm{B}} \times 1000}{6.0 \times\left(2500-\mathrm{W}_{\mathrm{B}}\right)}\)
Solving we get WB = 37.5 g.
![]()
Question 5.
Calculate
(a) molality
(b) molarity
(c) mole fraction of KI if the density of 20% (mass/ mass) aqueous KI is 1.202 g mL-1
Answer:
Mass of KI(WB) = 20 g
Molar mass of KI (MB) = 166 g mol-1
Density of solution (d) = 1.202 g mL-1
Mass of solution (WA + WB) = 100 g
Volume of solution (V) = \(\frac{\text { Mass }}{\text { density }}\)
= \(\frac{100}{1.202 \mathrm{gmL}^{-1}}\) = 83.2 ml
Mass of solvent WA = 100 – 20 = 80 g.
a) Molality (m) = \(\frac{W_B \times 1000}{M_B \times W_A}\)
= \(\frac{20 \mathrm{~g} \times 1000 \mathrm{~g} \mathrm{~kg}^{-1}}{166 \mathrm{~g} \mathrm{~mol}^{-1} \times 80 \mathrm{~g}}\)
= 1.51 mol kg-1
b) Molarity (M) = \(\frac{\mathrm{W}_{\mathrm{B}} \times 1000}{\mathrm{M}_{\mathrm{B}} \times \mathrm{V}}\)
= \(\frac{20 \mathrm{~g} \times 1000 \mathrm{~mL}}{166 \mathrm{~g} \mathrm{~mol}^{-1} \times 83.2 \mathrm{ml}}\)
= 1.45 mol L-1
c)
Moles of KI(nB) = \(\frac{20 \mathrm{~g}}{166 \mathrm{~g} \mathrm{~mol}^{-1}}\) = 1.51 mol
Moles of water (nA) = \(\frac{1000 \mathrm{~g}}{18 \mathrm{~g} \mathrm{~mol}^{-1}}\) = 55.5
∴ Mole fraction of KI (xB) = \(\frac{1.51}{1.51+55.5}\)
= 0.026
Question 6.
H2S, a toxic gas with rotten egg like smell, is used for qualitative analysis. If the solubility of H2S in water at STP is 0.195 m, calculate Henry’s law constant.
Answer:
According to Henry’s law
KH . x = p or KH = \(\frac{p}{x}\)
P = 1 bar, solubility = 0.195 mol kg-1
Moles of H2S (nB) = 0.195
Moles of water (nA) = \(\frac{1000 \mathrm{~g}}{18 \mathrm{~g} \mathrm{~mol}^{-1}}\) = 55.5
∴ Mole fraction of H2S (x)
= \(\frac{0.195}{0.195+55.5}\) = 3.5 × 10-3
KH = \(\frac{1(\text { bar) }}{3.5 \times 10^{-3}}\) = 285.6 bar
Question 7.
Henry’s law constant of CO2 in water is 1.67 × 108 Pa at 298K. Calculate the quantity of CO2 in 500 mL of soda water when packed under 25 atm CO2 pressure at 298 K.
Answer:
Pressure of CO2 (p) = 2.5 atm
= 2.5 atm × 101325\(\frac{(\mathrm{Pa})}{\mathrm{atm}}\)
= 253312.5 Pa
According to Henrys law
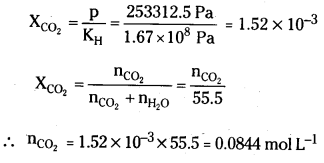
Moles of CO2 present in 500 ml
= \(\frac{0.0844}{2}\) mol = 0.0422 mol.
Amount of CO2 present in 500 ml
= 0.0422 mol × 440 g mol-1 = 1.86 g.
Question 8.
The vapour pressure of pure liquids A and B are 450 and 700 mm Hg respectively at 350K. Find out the composition of the liquid mixture if total vapour pressure is 600 mm Hg. Also find the composition of the vapour phase.
Answer:
Total vapour pressure PT = 600 mm Hg
Let Mole fraction of A is XA and that of B is XB
Vapour pressure of pure A(\(\mathrm{P}_{\mathrm{A}}^0\)) = 450 mm Hg
Vapour pressure of pure B(\(\mathrm{P}_{\mathrm{B}}^0\)) =700 mm Hg
Now PTotal = \(\mathrm{P}_{\mathrm{A}}^0 \mathrm{x}_{\mathrm{A}}\) + \(\mathrm{P}_{\mathrm{B}}^0 \mathrm{x}_{\mathrm{B}}\)
∴ 600 = 450 xA + 700 xB
= 450xA + 700(1 – xA)
= 700 – 250 xA or xA = 0.4
xB = 1 – xA = 1 – 0.4 = 0.6
Mole fraction of A in vapour phase
YA = \(\frac{x_A P_A^0}{P_{\text {Total }}^0}\) = \(\frac{0.4 \times 450}{600}\) = 0.3
Mole fraction of B in vapour phase
YB = \(\frac{\mathrm{x}_{\mathrm{B}} \mathrm{P}_{\mathrm{B}}^0}{\mathrm{P}_{\text {Total }}}\) = \(\frac{0.6 \times 700}{600}\) = 0.7
Question 9.
Vapour pressure of pure water at 298K is 23.8 mm Hg. 50 of urea (NH2 CO NH2) is dissolved in 850 g of water. Calculate the vapour pressure of water for this solution and its relative lowering.
Answer:
Mass of water = 850 g
Moles of water = \(\frac{850}{18}\) = 47.22 mol
Mass of urea = 50 g
Mol mass of urea = 60
Moles of urea = \(\frac{50}{60}\) = 0.83
Since the solution is not dilute the formula to be used is

Question 10.
Boiling point of water at 750 min Hg is 99.63°C. How much sucrose is to be added to 500g of water such that it boils at 100°C.
Answer:
Let mass of sucrose required = WB g
Mass of solvent WA = 500 g
Molar mass of sucrose (C12H22O11) = 342
Elevation in boiling point ΔTb = 100 – 99.63
= 0.37°C
ΔTb = \(\frac{\mathrm{K}_{\mathrm{b}} \times \mathrm{W}_{\mathrm{B}} \times 1000}{\mathrm{~W}_{\mathrm{A}} \times \mathrm{M}_{\mathrm{B}}}\)
(or) WB = \(\frac{\Delta \mathrm{T}_{\mathrm{b}} \times \mathrm{W}_{\mathrm{A}} \times \mathrm{M}_{\mathrm{B}}}{\mathrm{K}_{\mathrm{b}} \times 1000}\)
= \(\frac{0.37 \times 500 \times 342}{0.52 \times 1000}\) = 121.
Question 11.
Calculate the mass of ascorbic acid (vitamin C, C6H8O6) to be dissolved in 75g of
acetic acid to lower its melting point by 1.5°C. Kf = 3.9 K kg mol-1.
Answer:
Let the mass of ascorbic acid required = WBg
Molar mass of ascorbic acid (C6H8O6) MB = 176 g mol-1
Mass of solvent (WA) = 75 g
Depression in melting point ΔTf = 1.5°C
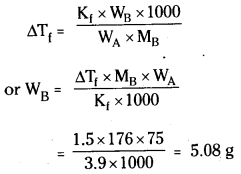
Question 12.
Calculate the osmotic pressure In pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 ion 450 ml of water at 37°C.
Answer:
Mass of polymer (WB) = 1.0 g
Molar mass of polymer MB = 1,85,000 g mol-1
T = 37 + 273.15 = 310.15K
π = \(\frac{W_B R_T}{M_B V_{(L)}}\)
= \(\frac{1 \times 0.082 \times 310.15}{185000 \times 450 \times 10^{-3}}\)
= 3.05 × 10-4 atm
= 3.05 × 10-4 × 1.01 × 105Pa = 30.9 Pa
Problems
Question 1.
Calculate the mole fraction of ethylene glycol (C2H6O2) in a solution containing 20% of C2H6O2 by mass. (TS 15)
Answer:
Assume that we have 100 g of solution (one can start with any amount of solution because the results obtained will be the same). Solution will contain 20 g of ethylene glycol and 80 g of water.
Molar mass of C2H6O2 = 12 × 2 + 1 × 6 + 16 × 2 = 62 g mol-1.

Question 2.
Calculate the molarity of a solution containing 5 g of NaOH in 450 mL solution. (TS Mar. 19; AP & TS 15)
Answer:
Moles of NaOH = \(\frac{5 \mathrm{~g}}{40 \mathrm{~g} \mathrm{~mol}^{-1}}\) = 0.125 mol
Volume of the solution in litres = 450 mL /1000 mL L-1
Using equation (2.8)
Molarity = \(\frac{0.125 \mathrm{~mol} \times 1000 \mathrm{~mL} \mathrm{~L}^{-1}}{450 \mathrm{~mL}}\)
= 0.278 M = 0.278 mol L-1
= 0.278 mol dm-3
Question 3.
Calculate molality of 2.5 g of ethanoic acid (CH3COOH) in 75 g of benzene. (TS 15)
Answer:
Molar mass of C2H4O2 = 12 × 2 + 1 × 4 + 16 × 2 = 60 g mol-1.
Moles mass of C2H4O2 = \(\frac{2.5 \mathrm{~g}}{60 \mathrm{~g} \mathrm{~mol}^{-1}}\)
= 0.0417 mol
Mass of benzene in kg = 75 g / 1000 g kg-1
= 75 × 10-3 kg
Molality of C2H4O2 = \(\frac{\text { moles of } \mathrm{C}_2 \mathrm{H}_4 \mathrm{O}_2}{\text { kg of benzene }}\)
= \(\frac{0.0417 \mathrm{~mol} \times 1000 \mathrm{~g} \mathrm{~kg}^{-1}}{75 \mathrm{~g}}\)
= 0.556 mol kg-1
![]()
Question 4.
If N2 gas is bubbled through water at 293K, how many mililmoles of N2 gas would dissolve in 1 litre of water? Assume that N2 exerts a partial pressure of 0.987 bar. Given that Henry’s law constant for N2 at 293 K is 76.48 kbar.
Answer:
The solubility of gas is related to the mole fraction in aqueous solution. The mole fraction of the gas in the solution is calculated by applying Henry’s law. Thus:
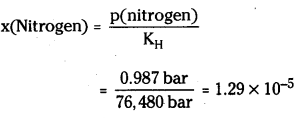
As 1 litre of water coñtains 55.5 mol of it, therefore If n represents number of moles of N2 in solution.
x(nitrogen) = \(\frac{n \mathrm{~mol}}{\mathrm{n} \mathrm{mol}+55.5 \mathrm{~mol}}\)
= \(\frac{\mathrm{n}}{55.5}\) = 1.29 × 10-5
(n in denominator is neglected as it is <<55.5)
Thus n = 1.29 × 10-5 × 55.5 mol
= 7.16 × 10-4 mol-1
= \(\frac{7.16 \times 10^{-4} \mathrm{~mol} \times 1000 \mathrm{~m} \mathrm{~mol}}{1 \mathrm{~mol}}\)
= 0.716 m mol
Question 5.
Vapour pressure of chloroform (CHCl3) and dichloromethane (CH2Cl2) at 298 K are 200mm Hg and 415 mm Hg respectively.
(i) Calculate the vapour pressure of the solution prepared by mixing 25.5 g of CHCl3 and 40 g of CH2Cl2 at 298 K and,
(ii) mole fractions of each component in vapour phase.
Answer:
i) Molar mass of CH2Cl2 = 12 × 1 + 1 × 2 + 35.5 × 2 = 85 g mol-1
Molar mass of CHCl3 = 12 × 1 + 1 × 1 + 35.5 × 3 = 119.5 g mol-1
Moles of CH2Cl2 = \(\frac{40 \mathrm{~g}}{85 \mathrm{~g} \mathrm{~mol}^{-1}}\) = 0.47 mol
Moles of CHCl3 = \(\frac{25.5 \mathrm{~g}}{119.5 \mathrm{~g} \mathrm{~mol}^{-1}}\)
= 0.213 mol
Total number of moles = 0.47 + 0.213
= 0.683 mol
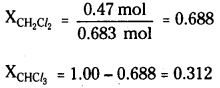
Using equation (2.16),
Ptotal = \(\mathrm{p}_1^0\) + (\(\mathrm{p}_2^0\) + \(\mathrm{p}_1^0\))x2
= 200 + (415 – 200) × 0.688
= 200 + 147.9 = 347.9 mm Hg
(ii) Using the relation (2.19) y1 = P1 / ptotal, we can calculate the mole fraction of the components in gas phase (y1).
\(\mathrm{p}_{\mathrm{CH}_2} \mathrm{Cl}_2\) = 0.688 × 415 mm Hg = 285.5 mm Hg
\(\mathbf{p}_{\mathrm{CHCl}_3}\) = 0.312 × 200 mm Hg = 62.4 mm Hg
\(\mathbf{y}_{\mathrm{CH}_2 \mathrm{Cl}_2}\) = 285.5 mm Hg / 347.9 mm Hg = 0.82
\(\mathbf{y}_{\mathrm{CHCl} 3}\) = 62.4 mm Hg/ 347.9 mm Hg = 0.18
Note : Since, CH2Cl2 is a more volatile component than CHCl3, [\(\mathrm{p}_{\mathrm{CH}_2}^0 \mathrm{Cl}_2\) = 415 mm Hg and \(\mathbf{p}_{\mathrm{CHC} l_3}^0\) = 200 mm Hg] and the vapour phase is also richer in CH2Cl2 [\(\mathbf{y}_{\mathrm{CH}_2} \mathrm{Cl}_2\) = 0.82 and \(\mathbf{y}_{\mathrm{CHCl}_3} \)= 0.18], it may thus be concluded that at equilibrium, vapour phase will be always rich in the component which is more volatile.
Question 6.
The vapour pressure of pure benzene at a certain temperature is 0.850 bar. A non – volatile, non – electrolyte solid weighing 0.5 g when added to 39.0 g of benzene (molar mass 78 g mol-1). Vapour pressure of the solution, then, is 0.845 bar. What is the molar mass of the solid substance ? (Mar. 2018, 16 AP)
Answer:
The various quantities known to us are as follows :
\(\mathrm{p}_1^0\) = 0.850 bar ; p = 0.845 bar
M1 = 78 g mol-1; w2 = 0.5 g; w1 = 39 g
Substituting these values in equation (2.28), we get
\(\frac{0.850 \mathrm{bar}-0.845 \mathrm{bar}}{0.850 \mathrm{bar}}\) = \(\frac{0.5 \mathrm{~g} \times 78 \mathrm{~g} \mathrm{~mol}^{-1}}{\mathrm{M}_2 \times 39 \mathrm{~g}}\)
Therefore, M2 = 170 g mol-1
Question 7.
18 g of glucose, C6H12O6, is dissolved in 1 kg of water In a saucepan. At what temperature will water boll at 1.013 bar? Kb for water is 0.52 K kg mol-1.
Answer:
Moles of glucose = 18 g / 180 g mol-1
= 0.1 mol
Number of kilograms of solvent = 1 kg
Thus molality of glucose solution
= 0.1 mol kg-1
For water, change in boiling point
ΔTb = Kb × m = 0.52 K kg mol-1 × 0.1 mol kg-1 = 0.052 K
Since water boils at 373.15 Kat 1.013 bar pressure, therefore, the boiling point of solution will be 373.15 + 0.052 = 373.202 K.
Question 8.
The boiling point of benzene is 353.23 K. When 1.80 g of a non – volatile solute was dissolved in 90 g of benzene, the boiling point is raised to 354.11 K. Calculate the molar mass of the solute. Kb for benzene is 2.53 K kg mol-1.
Answer:
The elevation (ΔTb) in the boiling point = 354.11 K – 352.23 K = 0.88 K
Substituting these values in expression (2.33) we get
M2 = \(\frac{2.53 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1} \times 1.8 \mathrm{~g} \times 1000 \mathrm{~g} \mathrm{~kg}^{-1}}{0.88 \mathrm{~K} \times 90 \mathrm{~g}}\)
= 58 g mol-1
Therefore, molar mass of the solute, M2 = 58 g mol-1
Question 9.
45 g of ethylene glycol (C2H6O2) is mixed with 600 g of water. Calculate
(a) the freezing point depression and
(b) the freezing point of the solution.
Answer:
Depression in freezing point is related to the molality, therefore, the molality of the solution with respect to ethylene glycol
moles of ethylene glycol mass of water in kilogram
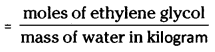
Moles of ethylene glycol = \(\frac{45 \mathrm{~g}}{62 \mathrm{~g} \mathrm{~mol}^{-1}}\)
= 0.73 mol
Mass of water in kg = \(\frac{600 \mathrm{~g}}{1000 \mathrm{~g} \mathrm{~kg}^{-1}}\) = 0.6 kg
Hence molality of ethylene glycol = \(\frac{0.73 \mathrm{~g}}{0.60 \mathrm{~kg}}\)
= 1.2 mol kg-1
Therefore freezing point depression.
ΔT1 = 1.86K kg mol-1 × 1.2 mol kg-1 = 2.2K
Freezing point of the aqueous solution
= 273.15 K – 2.2 K = 270.95 K
Question 10.
1.00 g of a non – electrolyte solute dissolved in 50 g of benzene lowered the freezing point of benzene by 0.40 K. The freezing
point depression constant of benzene is 5.12 K mol-1. Find the molar mass of the solute.
Answer:
Substituting the values of various terms involved in equation (2.36) we get.
M2 = \(\frac{5.12 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1} \times 1.00 \mathrm{~g} \times 1000 \mathrm{~g} \mathrm{~kg}^{-1}}{0.40 \times 50 \mathrm{~g}}\)
= 256 g mol-1
Thus, molar mass of the solute = 256 g mol-1
Question 11.
200 cm3 of an aqueous solution of a protein contains 1.26 g of the protein. The osmotic pressure of such a solution at 300 K is found to be 2.57 × 10-3 bar. Calculate the molar mass of the protein.
Answer:
The various quantities known to us are as follows : π = 2.57 × 10-3 bar,
V = 200 cm3 = 0.200 litre
T = 300 K
R = 0.083 L bar mol-1K-1
Substituting these values in equation (2.42) we get
M2 = \(\frac{1.26 \mathrm{~g} \times 0.083 \mathrm{~L} \mathrm{bar} \mathrm{K}^{-1} \mathrm{~mol}^{-1} \times 300 \mathrm{~K}}{2.57 \times 10^{-3} \mathrm{bar} \times 0.200 \mathrm{~L}}\)
= 61.022 g mol-1
Question 12.
2 g of benzoic acid (C6H5COOH) dissolved in 25 g of benzene shows a depression in freezing point equal 101.62K. Molal depression constant for benzene is 4.9 K kg mol-1. What is the percentage association of acid if it forms dimer in solution?
Answer:
The given quantities are : w2 = 2 g;
Kf = 4.9 K kg mol-1 ; w1 = 25 g,
ΔTf = 1.62 K
Substituting these values in equation (2.36) we get:
M2 = \(\frac{4.9 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1} \times 2 \mathrm{~g} \times 1000 \mathrm{~g} \mathrm{~kg}^{-1}}{25 \mathrm{~g} \times 1.62 \mathrm{~K}}\)
= 241.98 g mol-1
Thus, experimental molar mass of benzoic acid in benzene is = 241.98 g mol-1
Now consider the following equilibrium for the acid:
2C6H5COOH \(\rightleftharpoons\) (C6H5COOH)2
If x represents the degree of association of the solute then we would have (1 – x) mol of benzoic acid left in unassociated form and correspondingly \(\frac{x}{2}\) as associated moles of benzoic acid at equilibrium. Therefore, total number of moles of particles at equilibrium is :
1 – x + \(\frac{x}{2}\) = 1 – \(\frac{x}{2}\)
Thus, total number of moles of particles at equilibrium equals van’t Hoff factor i.

Therefore, degree of association of benzoic acid in benzene is 99.2%.
![]()
Question 13.
0.6 mL of acetic acid (CH3COOH), having density 1.06 g mL-1, is dissolved in 1 litre of water. The depression in freezing point observed for this strength of acid was 0.0205°C. Calculate the van’t Hoff factor and the dissociation constant of acid.
Answer:
Number of moles of acetic acid
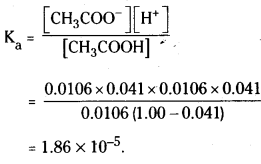
= \(\frac{0.6 \mathrm{~mL} \times 1.06 \mathrm{~g} \mathrm{~mL}^{-1}}{60 \mathrm{~g} \mathrm{~mol}^{-1}}\) = 0.0106 mol = n
Molarity = \(\frac{0.0106 \mathrm{~mol}}{1000 \mathrm{~mL} \times 1 \mathrm{~g} \mathrm{~mL}^{-1}}\)
= 0.0106 mol kg-1
Using equation (2.35)
∆Tf = 1.86 K kg mol-1 × 0.0106 mol kg-1 = 0.0197K
van’t Hoff factor

Acetic acid is a weak electrolyte and will dissociate into two ions : acetate and hydrogen ions per molecule of acetic acid. If x is the degree of dissociation of acetic acid, then we would have n (1 – x) moles of undisso-ciated acetic acid, nx moles of CH3COO– and nx moles of H+ ions.
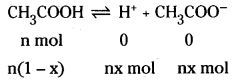
Thus total moles of particles are :
n(1 – x + x + x) = n(1 + x)
i = \(\frac{n(1+x)}{n}\) = 1 + x = 1.041
Thus degree of dissociation of acetic acid = x = 1.041 – 1.000 = 0.041
Then [CH3COOH] = n(1 – x)
= 0.0106 ( 1 – 0.041),
[CH3COO–] = nx = 0.0106 × 0.041, [H+]
= nx = 0.0106 × 0.041.