Telangana TSBIE TS Inter 2nd Year Physics Study Material 12th Lesson Dual Nature of Radiation and Matter Textbook Questions and Answers.
TS Inter 2nd Year Physics Study Material 12th Lesson Dual Nature of Radiation and Matter
Very Short Answer Type Questions
Question 1.
What are “Cathode rays”? [TS Mar. 19; AP mar. 1 7, may 18, 14]
Answer:
Cathode rays consists of a steam of fast moving negatively charged particles.
Speed of cathode rays ranges about 0.1 to 0.2 times light velocity (3 × 108m/s)
Question 2.
What important fact did Millikan’s experiment establish?
Answer:
Millikan proved the validity of Einstein’s photo electric equation and his successful explanation of photo electric effect using light quanta. He experimentally found the value of Planck’s constant ‘h’ and work function on ‘Φ’ of photo electric surface.
Question 3.
What is “work function”? [AP Mar. 19, May 16; TS Mar. 18. 17, 15]
Answer:
Work function (Φ) :
The mininum energy required by an electron to escape from metal surface is called “work function”.
Work function depends on nature of metal.
Question 4.
What is “photoelectric effect”? [AP Mar. ’18, ’17, ’15, ’14, May ’14; TS May ’17, Mar. ’18. ’16]
Answer:
Photoelectric effect :
The process of liberating an electron from the metal surface due to light energy falling on it is called “photoelectric effect”.
Question 5.
Give examples of “photosensitive substances”. Why are they called so?
Answer:
- Metals like zinc, cadmium and magnesium will respond to ultraviolet rays.
- Alkalimetals such as sodium, potassium, caesium and rubidium will respond to visible light.
These substances are called “photo sensitive surfaces” because they will emit electrons when light falls on them.
![]()
Question 6.
An electron, an a particle and a proton have the same kinetic energy. Which of these particles has the shortest de Broglie wavelength? [TS May, ’18, Mar. ’15]
Answer:
De-Broglie wavelength λ = \(\frac{h}{p}\); but KE = \(\frac{P^2}{2m}\)

For a particle m is more them given other particles.
So De-Broglie wavelength of a – particle is less.
Question 7.
What is Photo-electric effect? How did Einstein’s photo-electric equation explain the effects of intensity (of light) and potential on photo-electric current? [AP May 18, TS June 15]
Answer:
Photoelectric effect :
The process of emitting electron from the metal surface when light energy falling on it is called “photo-electrie effect”.
According to Einstein radiation consists of discrete units of energy called quanta of energy radiation.
Energy of quanta is called photon in light E = hυ
Maximum kinetic energy of photoelectron (Kmax) is the difference of energy of incident radiation (hυ) and work function (Φ)
∴ Kmax = hυ – Φ (when υ > υ0)
Photoelectric equation can also written as
![]()
Effect of intensity :
As per Einstein’s photoelectric equation energy of photon E = hυ decides weather a photon will come out of metal surface or not. If frequency of incident light υ > υ0 then electron will come out of that surface.
Number of electrons liberated depends on the number of photons striking the surface i.e., on intensity of light. So as per Einstein’s equation photocurrent liberated must be linearly proportional to intensity of light which is a practically proved fact.
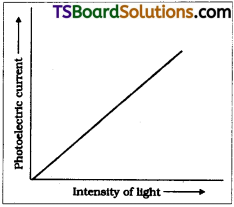
Effect of voltage on photocurrent :
When positive potential on collector is gradually increased then photocurrent i.e., also gradually increased upto certain limit called saturation current all the photoelectrons liberated from. Photosurface reached the collector.
When υ >υ0 photoelectron is released. The positive potential on collector will accelerate the electron. So it reaches the collector quickly. So photocurrent increases. But collector potential deals nothing with liberation of electron from photosurface.
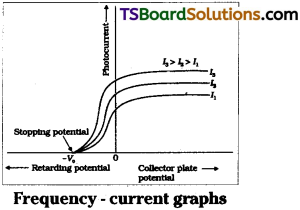
In this way Einstein’s photoelectric equation explained the saturation of photocurrent with increasing collector +ve potential. Effect of frequency and stopping potential.
From Einstein’s photoelectric equation
Kmax = hυ – Φ
i.e., kinetic energy of photoelectron is directly proportional to frequency of incident light.
Question 8.
Write down Einstein’s photoelectric equation. [AP Mar. 19, 15, May 17; TS May 18, 16]
Answer:
Maximum kinetic energy of photo electron Kmax is the difference of energy of incident radiation (hυ) and work function (Φ)
Kmax= hυ – Φ (when υ > υ0)
OR
Kmax = eV0 = hυ – Φ Or V0 = \(\frac{h}{e}\)υ – \(\frac{\phi_{0}}{e}\)
Question 9.
Write down de Broglie’s relation and explain the terms there in. [AP & TS Mar. 18, 16; TS May 17]
Answer:
de -Broglie assumed that matter will also exhibit wave nature when it is in motion.
de – Broglie wavelength, λ = \(\frac{h}{p}=\frac{h}{mυ}\)
P = mo, momentum of the body, h = Planck’s constant, λ = wave length of moving particle.
![]()
Question 10.
State Heisenberg’s uncertainty principle. [TS Mar. 17; AP Mar. 14]
Answer:
Heisenberg’s uncertainty principle :
We cannot exactly find both momentum and position of an electron at the same time. This is called Heisenberg’s uncertainty principle.
Short Answer Questions
Question 1.
What is the effect of (i) intensity of light (ii) potential on photoelectric current? [TS Mar. ’19, June ’15]
Answer:
Effect of intensity :
The number of photo electrons liberated is directly proportional to intensity of incident radiation. So photo current increases linearly with increase of intensity of incident light.
Effect of potential :
When positive potential given to collector, photocurrent is gradually increased up to certain limit called saturation current. In this stage all the photo electrons liberated from photo surface reached the collector.
When negative potential on collector is gradually increased electrons are repelled by collector and photo current decreases.
At a particular negative voltage photo current is zero.
Stopping potential :
The minimum negative potential required by collector to stop photo current (or) becomes zero is called cut-off voltage (V0) stopping potential.
Question 2.
How is the de-Broglie wavelength associated with an electron accelerated through a potential difference of 100 volts? [AP Mar. ’15]
Answer:
de-Broglie wavelength λ = \(\frac{h}{\sqrt{2 \mathrm{~m}} \mathrm{eV}}\)
Potential difference V = 100V; h = 6.63 × 10-34
Mass of electron m = 9.1 × 10-31 kg;
charge of electron e = 1.6 × 10-19c
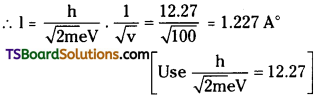
Question 3.
What is the de Broglie wavelength associated with an electron, accelerated through a potential difference of 100 volt? [TS May. ’16]
Answer:
Applied potential V = 100 V.
de Broglie wavelength
![]()
∴ de Broglie wavelength 1 = 0.1227 nm.
Question 4.
Describe an experiment to study the effect of frequency of incident radiation on ‘stopping potential’.
Answer:
Experimental study of photo electric effect :
To study photo electric effect a photo sensitive surface and a metallic plate called collector are arranged on an evacuated glass tube. An arrangement is made to give required positive or negative potential to collector. By changing the filters placed in the path of incident light we will allow light rays of required frequency to fall on given photo surface.
Stopping potential :
The minimum negative potential required by collector to stop photo current or photo current to become zero is called “cut off voltage V0“.
Effect of frequency on stopping potential are
- Stopping potential varies linearly with frequency of incident light.
- Every photo surface has a minimum cut off frequency υ0 for which stopping potential V0 = 0
![]()
Question 5.
Summarise the photon picture of electromagnetic radiation.
Answer:
Radiation consists of discrete units of energy called quanta.
Energy of quanta (E) = hυ = \(\frac{hc}{\lambda}\)
In case of light energy quanta is called photon.
Properties of photons:
- Energy of photon E = ho Momentum ho P= \(\frac{hυ}{e}\)
- In interaction of radiation with matter light quanta will behave like particles
- Photons are electrically neutral. So they are not deflected by electric and magnetic fields.
- In photons-particle collision total energy and total mometum of are conserved is in collision photon will totally loose its enery and momentum.
Question 6.
What is the deBroglie wavelength of a ball of mass 0.12Kg moving with a speed of 20ms-1? What can we infer from this result? [AP June ’15]
Answer:
Mass of ball, m = 0.12kg ;
Speed of ball, v = 20 m/s
Plancks’ constant, h = 6.63 × 10-34J
But de Broglie wave length λ = \(\frac{h}{p}=\frac{h}{mv}\)
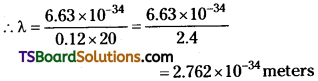
∴ de Broglie wave length of given moving ball X = 2.762 × 10-34m
Question 7.
The work function of cesium is 2.14 eV. Find the threshold frequency for cesium. (Take h = 6.6 × 10-34Js) [IMP]
Answer:
Work function Φ0 = 2.14 eV; h = 6.6 × 10-34 JS
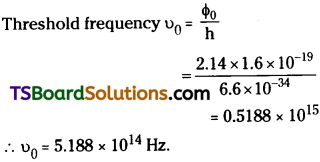
Long Answer Questions
Question 1.
How did Einstein’s photoelectric equation Explain the effect of intensity and potential on photoelectric current? How did this equation account for die effect of frequency of incident light on stopping potential?
Answer:
Einstein’s photo electric equation :
According to Einstein radiation consists of discrete units of energy called quanta of energy radiation.
Energy of quanta called photon in light E = hυ
Maximum kinetic energy of photo electron (Kmax) is the difference of energy of incident radiation (hυ) and work function (Φ)
∴ Kmax= hυ – Φ(when υ > υ0)
Photo electric equation can be written as
![]()
Effect of intensity :
As per Einstein’s photo electric equation energy of photon decides weather a photon will come out (or) not from metal surface. If frequency of incident light υ > υ0, then electron will come out from surface.
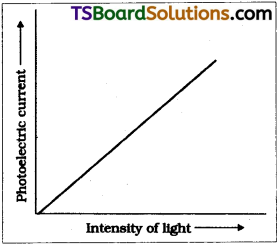
Number of electrons liberated depends on the number of photons striking the surface i.e., on intensity of light. So as per Einstein’s equation photo current liberated must be linearly proportional to intensity of light which is practically proved.
Effect of voltage on photo current :
When positive potential on collector is gradually increased then photo current i.e., also gradually increased up to certain limit called saturation current. In this stage all the photo electrons liberated from photo surface reached the collector.
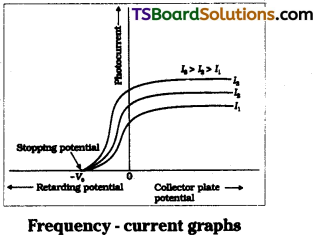
When υ > υ0 photo electron Is released. The positive potential on collector will accelerate the electron. So it reaches the collector quickly. So photo current increases. But collector potential deals nothing with liberation of electron from photo surface.
Effect of frequency and stopping potential :
⇒ Kinetic energy of photo electron is directly proportional to frequency of incident light.
From Einstein’s photo electric equation
∴ Kmax= hυ – Φ
Stopping potential :
The minimum negative potential required by the collector to stop photo current is called stopping potential.
At this potential even the fastest electron (or) electron with maximum kinetic energy is prevented to reach the collector.
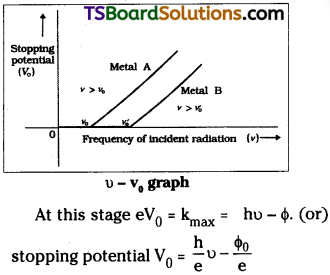
When frequency incident light increases then Kmax of electron increases. Hence stopping potential V0 will also increase.
∴ The graph between stopping potential V0 and frequency o must be a straight line.
Then slope of the line is \(\frac{h}{e}\). This is experimentally proved by Millikan.
In this way Einstein’s photo electric equation successfully explained photo electric effect.
![]()
Question 2.
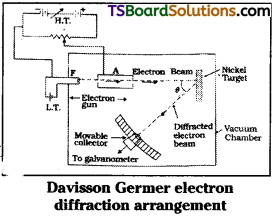
Describe the Davisson and Germer experiment. What did this experiment conclusively prove?
Answer:
Davisson and Germer experiment :
In this experiment, electrons are produced by heating a tungsten filament coated with barium oxide with the help of a low voltage battery.
These electrons are focussed on to a nickel target in the form of a sharp beam.
This electron beam is accelerated by the strong positive potential on nickel target.
After colliding the target electron beam will get scattered.
The scattered electron beam is collected by electron beam detector called collector.
In this experiment intensity of electron beam I for different angles of scattering is measured. A graph is plotted between angle of scattering 0 and intensity I with different target potentials of 44V to 68V.
Intensity is found to be maximum at a target potential of 54V with a scattering angle of 50°
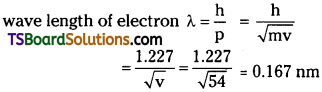
This value coincides with the de Broglie wave length of electron.
Importance :
This experiment proved the existence of matter waves practically. It gave a strong support to de Broglie’s hypothesis of matter wave concept.
Intext Question and Amswers
Question 1.
The photoelectric cut-off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
Answer:
Photoelectric cut-off voltage, V0 = 1.5 V ;
maximum kinetic energy Ke = eV0
Where, e = Charge on an electron = 1.6 × 10-19 C
∴ ke = 1.6 × 10-19 × 15 = 2.4 × 10-19J
∴ The maximum kinetic energy of the photoelectrons emitted K = 2.4 × 10-19 J.
Question 2.
The energy flux of sunlight reaching the surface of the earth is 1.388 × 10³ W/m². How many photons (nearly) per square metre are incident on the earth per second? Assume that the photons in the sunlight have an average wavelength of 550 nm.
Answer:
Energy flux of sunlight reaching the surface of earth, Φ = 1.388 × 10³ W/m²
Hence, power of sunlight per square metre, P = 1.388 × 10³W
Speed of light, c = 3 × 108 m/s ; Planck’s constant, h = 6.626 × 10-34 Js
Average wavelength of photons present in sunlight, λ = 550 nm = 550 × 10-19 m
Number of photons per square metre incident on earth per second = n
∴ Power p = n E
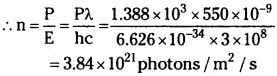
∴ 3.84 × 1021 photons are incident per square metre / sec on earth.
Question 3.
In an experiment on photoelectric effect, the slope of the cut-off voltage versus frequency of incident light is found to be 4.12 × 10-15 V s. Calculate the value of Planck’s constant.
Answer:
The slope of the cut-off voltage (V) – frequency (v) graph is ; \(\frac{V}{v}\) = 4.12 × 10-15 Vs
But hv = eV
Where, e = Charge on an electron = 1.6 × 10-19 C; h = Planck’s constant
∴ h = e×\(\frac{V}{v}\) = 1.6 × 10-19 × 4.12 × 10-15
= 6.592 × 10-34Js
∴ Planck’s constant h = 6.592 × 10-34 Js.
Question 4.
A 100 W sodium lamp radiates energy uniformly in all directions. The lamp is located at the centre of a large sphere that absorbs all the sodium light which is incident on it. The wavelength of the sodium light is 589 nm. (a) What is the energy per photon associated with the sodium light? (b) At what rate are the photons delivered to the sphere?
Answer:
Power of the sodium lamp, P = 100 W ;
Wavelength of the emitted sodium light,
λ = 589 nm = 589 × 10-9 m
Planck’s constant, h = 6.626 × 10-34 Js ;
Speed of light, c = 3 × 108 m/s
(a) Energy of photon E = \(\frac{hc}{\lambda}\)
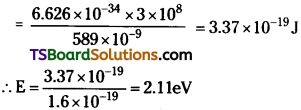
(b) Number of photons delivered to the sphere = n
Power P = nE
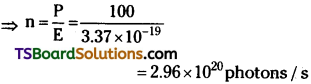
Photons delivered per second = 2.96 × 1020
Question 5.
The threshold frequency for a certain metal is 3.3 × 1014 Hz. If light of frequency 8.2 × 1014 Hz is incident on the metal, predict the cut-off voltage for the photoelectric emission.
Answer:
Threshold frequency of the metal,
v0 = 3.3 × 1014 Hz;
Charge on an electron, e = 1.6 × 10-19 C ;
Frequency of light incident on the metal, v0 = 8.2 × 1014 HZ;
Planck’s constant, h = 6.626 × 10-34 Js
Cut-off voltage for the photoelectric emission from the metal = V0; But eV0 = h(υ – υ0)

∴ The cut-off voltage for the photoelectric emission V0 = 2.0292 V.
![]()
Question 6.
The work function for a certain metal is 4.2 eV. Will this metal give photoelectric emission for incident radiation of wave-length 330 nm?
Answer:
No.
Work function of the metal, Φ0 = 4.2 eV;
Charge on an electron, e = 1.6 × 10-19 C
Planck’s constant, h = 6.626 × 10-34 Js ;
Wavelength of the incident radiation,
λ = 330 nm = 330 × 10-9 m
Speed of light, c = 3 × 108 m/s ;
The energy of the incident photon E = \(\frac{hc}{\lambda}\)

Question 7.
light of frequency 7.21 × 1014 Hz is incident on a metal surface. Electrons with a maximum speed of 6.0 × 105 m/s are ejected from the surface. What is the threshold frequency for photoemission of electrons?
Answer:
Frequency of photon, v = 488 nm
= 488 × 10-9 m ;
Planck’s constant, h = 6.626 × 10-34 Js
Maximum speed of the electrons, v = 6.0 × 105 m/s ;
Mass of an electron, m = 9.1 × 10-31 kg
Relation between v and K.E, \(\frac{1}{2}\) mv²
= h(ν – ν0)⇒ ν0 = ν –\(\frac{mv^2}{2h}\)
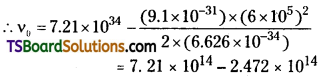
Threshold frequency ν0 = 4.738 × 105 Hz
Question 8.
Light of wavelength 488 nm is produced by an argon laser which is used in the photoelectric effect. When light from this spectral line is incident on the emitter, the stopping (cut-off) potential of photoelectrons is 0.38 V. Find the work function of the material from which the emitter is made.
Answer:
Wavelength of light produced by the argon laser, λ = 488 nm = 488 × 10-9 m
Stopping potential, V0 = 0.38 V ;
But leV = 1.6 × 10-19 J
∴ V0 = \(\frac{0.38}{1.6\times10^{-19}}\)eV
Planck’s constant, h = 6.6 × 10-34 Js ;
Charge on an electron, e = 1.6 × 10-19 C
Speed of light, c = 3 × 10 m/s
From Einstein’s photoelectric effect,

Question 9.
Calculate the
(a) Momentum/and
(b) De Broglie wavelength of the electrons accelerated through a potential difference of 56 V.
Answer:
Potential difference, V = 56 V;
Planck’s constant, h = 6.6 × 10-34 Js
Mass of an electron, m = 9.1 × 10-31 kg ;
Charge on an electron, e = 1.6 × 10-19 C
(a) At equilibrium, the kinetic energy of each electron is equal to the accelerating potential, for

The momentum of each accelerated electron
p = mv = 9.1 × 10-31 × 4.44 × 106
Momentum of each electron p
= 4.04 × 10-24 kg m s-1
(b)De Broglie wavelength of an electron,

Question 10.
What is the
(a) Momentum,
(b) Speed, and
(c) De Broglie wavelength of an electron with kinetic energy of 120 eV.
Answer:
Kinetic energy of the electron, Ek = 120 eV;
Planck’s constant, h = 6.6 × 10-34 Js
Mass of an electron, m = 9.1 × 10-31 kg ;
Charge on an electron, e = 1.6 × 10-19 C
(a) Kinetic energy of electron Ek = \(\frac{1}{2}\)mv²
Where, υ = Speed of the electron
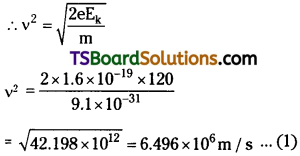
Momentum of the electron, p = mv
∴ P = 9.1 × 10-31 × 6.496 × 106
= 5.91 × 10-24 kg m s-1.
(b) Speed of the electron, v = 6.496 × 106 m/s (from eq 1)
(c) De Broglie wavelength of an electron having a momentum p, is given as:
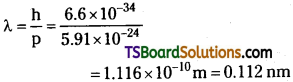
∴ de Broglie wavelength of the electron is 0.112 nm.
Question 11.
What is the de Broglie wavelength of
(a) a bullet of mass 0.040 kg travelling at the speed of 1.0 km/s,
(b) a ball of mass 0.060 kg moving at a speed of 1.0 m/s, and
(c) a dust particle of mass 1.0 × 10-9 kg drifting with a speed of 2.2 m/s?
Answer:
(a) Mass of the bullet, m = 0.040 kg ;
Speed of the bullet, v = 1.0 km/s
= 1000 m/s
Planck’s constant, h = 6.6 × 10-34Js
But De Broglie wavelength λ = \(\frac{h}{mv}\)
![]()
(b)Mass of the ball, m = 0.060 kg
Speed of the ball, v = 1.0 m/s
De Broglie wavelength λ = \(\frac{h}{mv}\)
![]()
(c) Mass of the dust particle, m =1 × 10-9 kg;
Speed of the dust particle, v = 2.2 m/s
De Broglie wavelength λ = \(\frac{h}{mv}\)
![]()
![]()
Question 12.
An electron and a photon each have a wavelength of 1.00 nm. Find
(a) their momenta,
(b) the energy of the photon, and
(c) the kinetic energy of electron.
Answer:
Wavelength of an electron (λe) and a photon (λp), λe = λp = λ = 1nm = 1 × 10-9 m
Planck’s constant, h = 6.63 × 10-34 Js
(a) The momentum of an elementary partide is given by de Broglie relation:
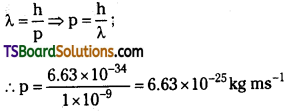
(b) The energy of a photon is given by the relation:

(c) The kinetic energy (A) of an electron having momentum p,is given by the relation:
m = Mass of the electron = 9.1 × 10-31 kg;
p = 6.63 × 10-25 kg ms-1
