Here students can locate TS Inter 2nd Year Physics Notes 14th Lesson Nuclei to prepare for their exam.
TS Inter 2nd Year Physics Notes 14th Lesson Nuclei
→ Nearly 99.9% of mass of atom is concentrated in a small volume called Nucleus.
→ Radius of atom is nearly 10,000 times more than radius of nucleus.
→ Volume of nucleus is nearly 10-12 times less than volume of atom.
→ Atomic mass unit (1u): 1 /12th mass of 126C atom is taken as “atomic mass unit”.
1.u = \(\frac{1.992647 \times 10^{-26}}{12}\) = 1.660539 x 10-27
Energy equivalent of 1u = 931.5 MeV.
→ Isotopes: The nuclei having the same atomic number (Z) but different mass number (A) are called “isotopes”. Ex: 8O16, 8O17, 8O18.
→ Isobars: The nuclei having the same mass number (A) but different atomic numbers (Z) are called “isobars”. Ex: 146C, 147N
→ Isotones : The nuclei having same neutron number (N) but different atomic number (Z) are called “isotones”. Ex: 80Hg198, 79197Au.
→ Isomers : Nuclei having the same atomic number (Z) and mass number (A) but with different nuclear properties such as radio-active decay and magnetic moments are called “isomers”.
Ex: I3580 Brm, 8035Brg . Here ‘m’ denotes metastable state and ‘g’ denotes ground state.
→ Positive charge of nucleus is due to protons.
→ Toted charge of electrons in an atom is (- Ze) and that of protons is (+ Ze). Where Z is atomic number.
→ Neutron is a chargeless particle. Mass of neutron and mass of proton are almost equal.
![]()
→ A free neutron is unstable when it is outside the nucleus. Its mean life period is 1000 sec.
→ Inside nucleus neutron is stable.
→ Number of neutrons in an atom is (A – Z) where A is mass number and Z is “atomic number”.
→ Volume of nucleus is proportional to mass number V ∝ A (OR) \(\frac{4}{3}\)πR3 ∝ A ⇒ R = R0A1/3 where RQ is a constant. R0 = 1.2 × 10-15 m.
→ Density of nuclear matter is almost cons-tant. It is independent of mass number A.
→ Density of nuclear matter ρn = 2.3 × 1017 kg/m3.
→ Einstein mass .energy equation: From theory of relativity mass is treated as another form of energy. Relation between mass and energy is E = mc2. Where c = Velocity of light = 3 × 108 m/s.
→ In a nuclear reaction Law of conservation of energy states that the initial energy and, final energy are equal provided the energy associated with mass is also taken into account.
→ Mass defect: In every nucleus the theore-tical mass (MT) is always less than practical mass (M). The difference of mass of nucleus and its constituents is known as “mass defect”. Mass defect Δm = [Zmp + (A – Z) mn] – M.
→ Binding energy : When a certain number of protons and neutrons are brought together to form a nucleus the certain amount of energy Eb is released.
The energy released while forming a nucleus is called “Binding energy Eb“. Binding energy = Δmc2.
Note : We have to supply an amount of energy equals to Eb from outside to divide a nucleus into its constituents.
→ Nuclear force:
- A nuclear force is much stronger than the coulomb1 force between the charges or the gravitational force between masses.
- Nuclear force between two nucleons is distance dependent.
- From potential energy graph of a pair of nucleons these forces are found to be attractive forces when separation between nucleons is 0.8 Fermi or more. These forces are found to be repulsive forces when separation between nucleons is less than 0.8 Fermi.
- Nuclear forces are saturated forces.
- Nuclear forces does not depend on charge. So nuclear force between proton-proton, proton – neutron and neutron – neutron are equal.
→ Radioactive decay : The spontaneous disintegration of unstable nucleus is referred as “radioactivity or radioactive decay”.
When a nucleus undergoes radioactive decay three types of radioactive decay takes place.
- α – decay : In this process 24He nuclei are emitted.
- β – deay : In this process electrons or positrons are emitted.
- γ – decay : In this process high energy photons (E.M. Waves) are liberated.
→ Law of radioactive decay : Let N is the number of nuclei in a sample. The number of nuclei (ΔN) undergoing radioactive decay during the time ‘Δt’ is given by
\(\frac{\Delta \mathrm{N}}{\Delta \mathrm{t}}\) N or \(\frac{\Delta \mathrm{N}}{\Delta \mathrm{t}}\) = λN
Where λ is disintegration constant or decay constant.
→ Decay rate (R) or Activity: The total decay rate of a sample is the number of nuclei disintegrating per unit time.
∴ Total decay rate R = – \(\frac{\mathrm{dN}}{\mathrm{dt}}\) (OR)
R = R0eλt (or) R = λN (activity )
Total decay rate is also called activity.
![]()
→ Half – life period (T1/2): The half-life period of a radioactive nuclide is the time taken for the number of nuclei (N) to become half of initial nuclei (No) i.e., N = \(\frac{\mathrm{N}_{\mathrm{o}}}{2}\).
→ Average life time : In a radioactive substance some nuclei may live for a long time and some nuclei may live for a short time. So we are using average life time T.
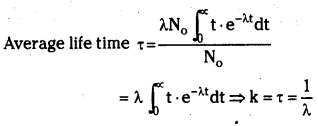
→ Becquerel (Bq): Becquerel is a unit to mea-sure radioactivity of a substance.
If a radioactive substance ungergoes 1 disintegration or decay per second then it is called Becquerel.
→ Curie: It is a unit to measure radioactivity of a substance.
If a radioactive substance undergoes 3.7 × 1010 decays per second then radioactivity of that substance is called curie.
Note:
- Curie is a very big unit. So generally millicurie is used to measure radioactivity,
- 1 Curie = 3.7 × 1010 Bq (Becquerel)
→ Alpha decay : In α – decay 2He4 nuclie is emitted from given radioactive substance. So mass number of product nucleus (called daughter nucleus) is decreased by four units and atomic number is decreased by two units. Equation of α – decay is
AZX → A-4Z-2X + 42He (a-particle)
→ Average life time τ = \(\frac{\mathrm{T}}{0.693}=\frac{\text { Half }-\text { life period }}{0.693}\)
→ Power of nuclear reactor P = \(\frac{\text { Number of fission } \times \text { Energy per fission }}{\text { time }}\)
Or
P = \(\frac{n}{t}\) × E